We now study the relation between the frequency ν of the incident radiation and the stopping potential V0. We suitably adjust the same intensity of light radiation at various frequencies and study the variation of photocurrent with collector plate potential. The resulting variation is shown in Fig. 11.4. We obtain different values of stopping potential but the same value of the saturation current for incident radiation of different frequencies. The energy of the emitted electrons depends on the frequency of the incident radiations. The stopping potential is more negative for higher frequencies of incident radiation. Note from Fig. 11.4 that the stopping potentials are in the order V03 > V02 > V01 if the frequencies are in the order ν3 > ν2 > ν1 . This implies that greater the frequency of incident light, greater is the maximum kinetic energy of the photoelectrons. Consequently, we need greater retarding potential to stop them completely. If we plot a graph between the frequency of incident radiation and the corresponding stopping potential for different metals we get a straight line, as shown in Fig. 11.5.
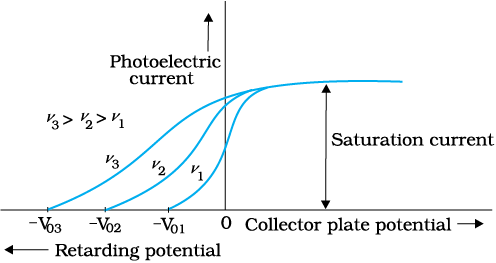
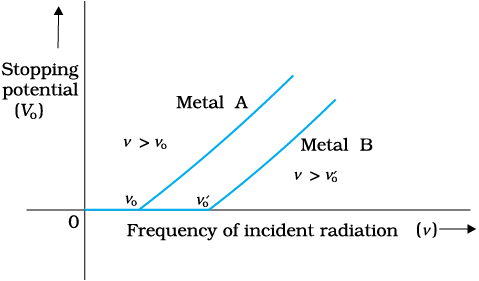
Figure 11.5 Variation of stopping potential V0 with frequency ν of incident radiation for a given photosensitive material.
The graph shows that
(i) the stopping potential V0 varies linearly with the frequency of incident radiation for a given photosensitive material.
(ii) there exists a certain minimum cut-off frequency ν0 for which the stopping potential is zero.
These observations have two implications:
(i) The maximum kinetic energy of the photoelectrons varies linearly with the frequency of incident radiation, but is independent of its intensity.
(ii) For a frequency ν of incident radiation, lower than the cut-off frequency ν0, no photoelectric emission is possible even if the intensity is large.
This minimum, cut-off frequency ν0, is called the threshold frequency. It is different for different metals.
Different photosensitive materials respond differently to light. Selenium is more sensitive than zinc or copper. The same photosensitive substance gives different response to light of different wavelengths. For example, ultraviolet light gives rise to photoelectric effect in copper while green or red light does not.
Note that in all the above experiments, it is found that, if frequency of the incident radiation exceeds the threshold frequency, the photoelectric emission starts instantaneously without any apparent time lag, even if the incident radiation is very dim. It is now known that emission starts in a time of the order of 10–9 s or less.
We now summarise the experimental features and observations described in this section.
(i) For a given photosensitive material and frequency of incident radiation (above the threshold frequency), the photoelectric current is directly proportional to the intensity of incident light (Fig. 11.2).
(ii) For a given photosensitive material and frequency of incident radiation, saturation current is found to be proportional to the intensity of incident radiation whereas the stopping potential is independent of its intensity (Fig. 11.3).
(iii) For a given photosensitive material, there exists a certain minimum cut-off frequency of the incident radiation, called the threshold frequency, below which no emission of photoelectrons takes place, no matter how intense the incident light is. Above the threshold frequency, the stopping potential or equivalently the maximum kinetic energy of the emitted photoelectrons increases linearly with the frequency of the incident radiation, but is independent of its intensity (Fig. 11.5).
(iv) The photoelectric emission is an instantaneous process without any apparent time lag (∼10–9s or less), even when the incident radiation is made exceedingly dim.