Optical isomers are mirror images that cannot be superimposed on one another. These are called as enantiomers. The molecules or ions that cannot be superimposed are called chiral. The two forms are called dextro (d) and laevo (l) depending upon the direction they rotate the plane of polarised light in a polarimeter (d rotates to the right, l to the left). Optical isomerism is common in octahedral complexes involving didentate ligands (Fig. 9.6).
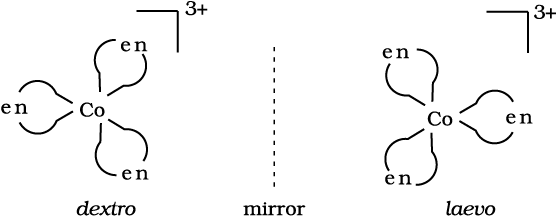
Fig.9.6: Optical isomers (d and l) of [Co(en)3] 3+
In a coordination entity of the type [PtCl2(en)2]2+, only the cis-isomer shows optical activity (Fig. 9.7).
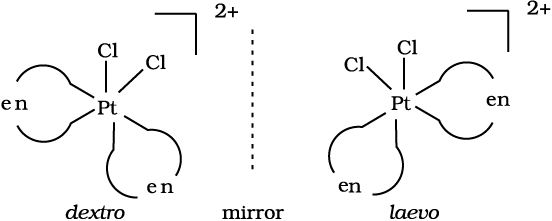
Fig. 9.7 Optical isomers (d and l) of cis-[PtCl2(en)2]2+
Draw structures of geometrical isomers of [Fe(NH3)2(CN)4]-
Solution
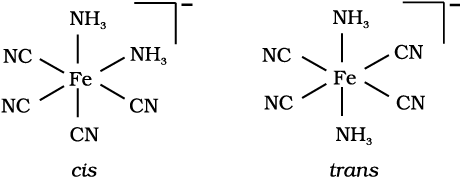
Example 9.6
Out of the following two coordination entities which is chiral (optically active)?
(a) cis-[CrCl2(ox)2]3– (b) trans-[CrCl2(ox)2]3–
Solution
The two entities are represented as
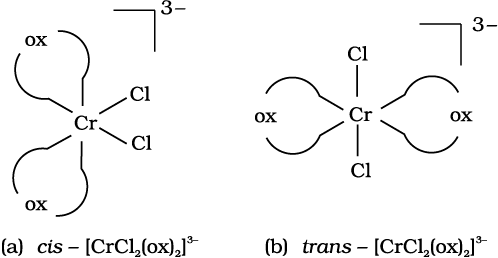
Out of the two, (a) cis - [CrCl2(ox)2]3- is chiral (optically active).

© 2026 GoodEd Technologies Pvt. Ltd.