This type of isomerism arises in heteroleptic complexes due to different possible geometric arrangements of the ligands. Important examples of this behaviour are found with coordination numbers 4 and 6. In a square planar complex of formula [MX2L2] (X and L are unidentate), the two ligands X may be arranged adjacent to each other in a cis isomer, or opposite to each other in a trans isomer as depicted in Fig. 9.2.
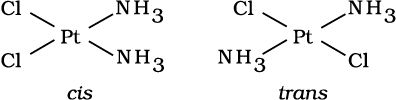
Fig. 9.2: Geometrical isomers (cis and trans) of Pt [NH3)2Cl2]
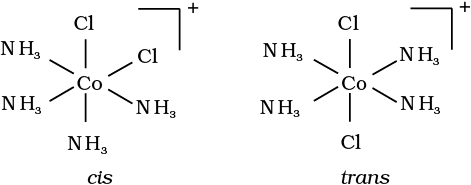
Fig. 9.3: Geometrical isomers (cis and trans) of [Co(NH3)4Cl2]+
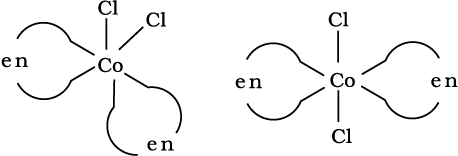
Fig. 9.4: Geometrical isomers (cis and trans) of [CoCl2(en)2]
Another type of geometrical isomerism occurs in octahedral coordination entities of the type [Ma3b3] like [Co(NH3)3(NO2)3]. If three donor atoms of the same ligands occupy adjacent positions at the corners of an octahedral face, we have the facial (fac) isomer. When the positions are around the meridian of the octahedron, we get the meridional (mer) isomer (Fig. 9.5).
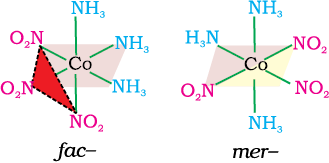
Fig. 9.5 The facial (fac) and meridional (mer) isomers of [Co(NH3)3(NO2)3]
Why is geometrical isomerism not possible in tetrahedral complexes having two different types of unidentate ligands coordinated with the central metal ion ?
Solution
Tetrahedral complexes do not show geometrical isomerism because the relative positions of the unidentate ligands attached to the central metal atom are the same with respect to each other.

© 2026 GoodEd Technologies Pvt. Ltd.