Oxidation states and trends in chemical reactivity
In the case of nitrogen, all oxidation states from +1 to +4 tend to disproportionate in acid solution. For example,
3HNO2 → HNO3 + H2O + 2NO
Similarly, in case of phosphorus nearly all intermediate oxidation states disproportionate into +5 and –3 both in alkali and acid. However +3 oxidation state in case of arsenic, antimony and bismuth becomes increasingly stable with respect to disproportionation.
Nitrogen is restricted to a maximum covalency of 4 since only four (one s and three p) orbitals are available for bonding. The heavier elements have vacant d orbitals in the outermost shell which can be used for bonding (covalency) and hence, expand their covalence as in PF–6.
Anomalous properties of nitrogen
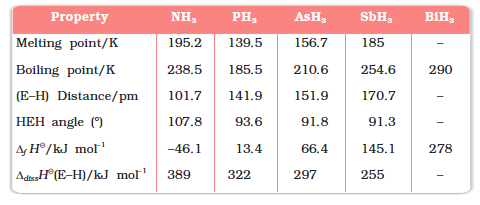
(i) Reactivity towards hydrogen: All the elements of Group 15 form hydrides of the type EH3 where E = N, P, As, Sb or Bi. Some of the properties of these hydrides are shown in Table 7.2. The hydrides show regular gradation in their properties. The stability of hydrides decreases from NH3 to BiH3 which can be observed from their bond dissociation enthalpy. Consequently, the reducing character of the hydrides increases. Ammonia is only a mild reducing agent while BiH3 is the strongest reducing agent amongst all the hydrides. Basicity also decreases in the order NH3 > PH3 > AsH3 > SbH3 > BiH3. Due to high electronegativity and small size of nitrogen, NH3 exhibits hydrogen bonding in solid as well as liquid state. Because of this, it has higher melting and boiling points than that of PH3.
Table 7.2: Properties of Hydrides of Group 15 Elements
(ii) Reactivity towards oxygen: All these elements form two types of oxides: E2O3 and E2O5. The oxide in the higher oxidation state of the element is more acidic than that of lower oxidation state. Their acidic character decreases down the group. The oxides of the type E2O3 of nitrogen and phosphorus are purely acidic, that of arsenic and antimony amphoteric and those of bismuth predominantly basic.
(iii) Reactivity towards halogens: These elements react to form two series of halides: EX3 and EX5. Nitrogen does not form pentahalide due to non-availability of the d orbitals in its valence shell. Pentahalides are more covalent than trihalides. This is due to the fact that in pentahalides +5 oxidation state exists while in the case of trihalides +3 oxidation state exists. Since elements in +5 oxidation state will have more polarising power than in +3 oxidation state, the covalent character of bonds is more in pentahalides. All the trihalides of these elements except those of nitrogen are stable. In case of nitrogen, only NF3 is known to be stable. Trihalides except BiF3 are predominantly covalent in nature.
Example 7.1
Though nitrogen exhibits +5 oxidation state, it does not form pentahalide. Give reason.
Example 7.2
PH3 has lower boiling point than NH3. Why?
Solution
Unlike NH3, PH3 molecules are not associated through hydrogen bonding in liquid state. That is why the boiling point of PH3 is lower than NH3.
Intext Questions
7.1 Why are pentahalides of P, As, Sb and Bi more covalent than their trihalides?
7.2 Why is BiH3 the strongest reducing agent amongst all the hydrides of Group 15 elements ?