A catalyst is a substance which increases the rate of a reaction without itself undergoing any permanent chemical change. For example, MnO2 catalyses the following reaction so as to increase its rate considerably.
2KClO3 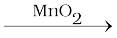 2 KCl + 3O2
2 KCl + 3O2
The word catalyst should not be used when the added substance reduces the rate of raction. The substance is then called inhibitor. The action of the catalyst can be explained by intermediate complex theory. According to this theory, a catalyst participates in a chemical reaction by forming temporary bonds with the reactants resulting in an intermediate complex. This has a transitory existence and decomposes to yield products and the catalyst.
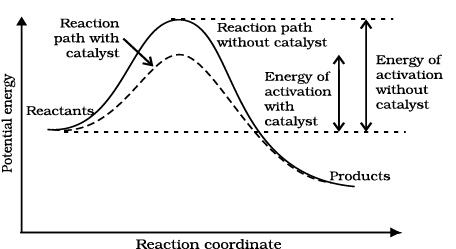
It is clear from Arrhenius equation (4.18) that lower the value of activation energy faster will be the rate of a reaction.
A small amount of the catalyst can catalyse a large amount of reactants. A catalyst does not alter Gibbs energy, ∆G of a reaction. It catalyses the spontaneous reactions but does not catalyse non-spontaneous reactions. It is also found that a catalyst does not change the equilibrium constant of a reaction rather, it helps in attaining the equilibrium faster, that is, it catalyses the forward as well as the backward reactions to the same extent so that the equilibrium state remains same but is reached earlier.

© 2026 GoodEd Technologies Pvt. Ltd.