Molecules in a gas have rather large speeds of the order of the speed of sound. Yet a gas leaking from a cylinder in a kitchen takes considerable time to diffuse to the other corners of the room. The top of a cloud of smoke holds together for hours. This happens because molecules in a gas have a finite though small size, so they are bound to undergo collisions. As a result, they cannot move straight unhindered; their paths keep getting incessantly deflected.
Seeing is Believing
Can one see atoms rushing about. Almost but not quite. One can see pollen grains of a flower being pushed around by molecules of water. The size of the grain is ~ 10-5 m. In 1827, a Scottish botanist Robert Brown, while examining, under a microscope, pollen grains of a flower suspended in water noticed that they continuously moved about in a zigzag, random fashion.
Kinetic theory provides a simple explanation of the phenomenon. Any object suspended in water is continuously bombarded from all sides by the water molecules. Since the motion of molecules is random, the number of molecules hitting the object in any direction is about the same as the number hitting in the opposite direction. The small difference between these molecular hits is negligible compared to the total number of hits for an object of ordinary size, and we do not notice any movement of the object.
When the object is sufficiently small but still visible under a microscope, the difference in molecular hits from different directions is not altogether negligible, i.e. the impulses and the torques given to the suspended object through continuous bombardment by the molecules of the medium (water or some other fluid) do not exactly sum to zero. There is a net impulse and torque in this or that direction. The suspended object thus, moves about in a zigzag manner and tumbles about randomly. This motion called now ‘Brownian motion’ is a visible proof of molecular activity. In the last 50 years or so molecules have been seen by scanning tunneling and other special microscopes.
In 1987 Ahmed Zewail, an Egyptian scientist working in USA was able to observe not only the molecules but also their detailed interactions. He did this by illuminating them with flashes of laser light for very short durations, of the order of tens of femtoseconds and photographing them. ( 1 femto- second = 10-15 s ). One could study even the formation and breaking of chemical bonds. That is really seeing !
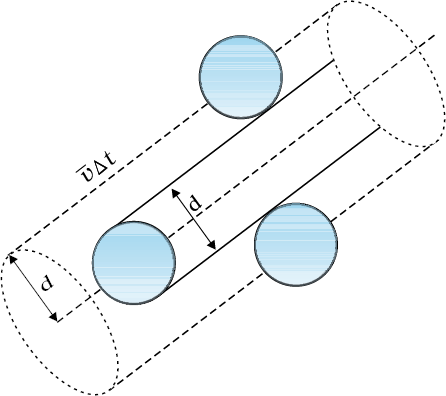
Suppose the molecules of a gas are spheres of diameter d. Focus on a single molecule with the average speed <v>. It will suffer collision with any molecule that comes within a distance d between the centres. In time ∆t, it sweeps a volume πd2 <v> ∆t wherein any other molecule will collide with it (see Fig. 13.7). If n is the number of molecules per unit volume, the molecule suffers nπd2 <v> ∆t collisions in time ∆t. Thus the rate of collisions is nπd2 <v> or the time between two successive collisions is on the average,
τ = 1/(nπ <v> d2 ) ..........(13.38)
The average distance between two successive collisions, called the mean free path l, is :
l = <v> /τ = 1/(nπd2) ............(13.39)
In this derivation, we imagined the other molecules to be at rest. But actually all molecules are moving and the collision rate is determined by the average relative velocity of the molecules. Thus we need to replace <v> by <vr> in Eq. (13.38). A more exact treatment gives
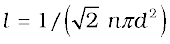 .............(13.40)
.............(13.40)
Let us estimate l and τ for air molecules with average speeds <v> = ( 485m/s). At STP
n = 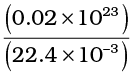
= 2.7 × 10 25 m -3.
Taking, d = 2 × 10–10 m,
τ = 6.1 × 10–10 s
and l = 2.9 × 10–7 m ≈ 1500d ..............(13.41)
As expected, the mean free path given by Eq. (13.40) depends inversely on the number density and the size of the molecules. In a highly evacuated tube n is rather small and the mean free path can be as large as the length of the tube.
Example 13.9 Estimate the mean free path for a water molecule in water vapour at 373 K. Use information from Exercises 13.1 and Eq. (13.41) above.
Answer The d for water vapour is same as that of air. The number density is inversely proportional to absolute temperature.
So 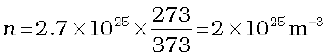
Hence, mean free path 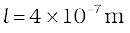
Note that the mean free path is 100 times the interatomic distance ~ 40 Å = 4 ×10-9 m calculated earlier. It is this large value of mean free path that leads to the typical gaseous behaviour. Gases can not be confined without a container.
Using, the kinetic theory of gases, the bulk measurable properties like viscosity, heat conductivity and diffusion can be related to the microscopic parameters like molecular size. It is through such relations that the molecular sizes were first estimated.