These hydrocarbons are also known as ‘arenes’. Since most of them possess pleasant odour (Greek; aroma meaning pleasant smelling), the class of compounds was named as ‘aromatic compounds’. Most of such compounds were found to contain benzene ring. Benzene ring is highly unsaturated but in a majority of reactions of aromatic compounds, the unsaturation of benzene ring is retained. However, there are examples of aromatic hydrocarbons which do not contain a benzene ring but instead contain other highly unsaturated ring. Aromatic compounds containing benzene ring are known as benzenoids and those not containing a benzene ring are known as non-benzenoids. Some examples of arenes are given
below:
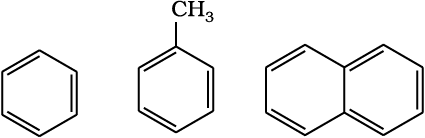
Benzene Toluene Naphthalene
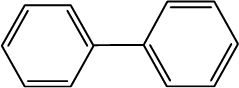
Biphenyl
The nomenclature and isomerism of aromatic hydrocarbons has already been discussed in Unit 12. All six hydrogen atoms in benzene are equivalent; so it forms one and only one type of monosubstituted product. When two hydrogen atoms in benzene are replaced by two similar or different monovalent atoms or groups, three different position isomers are possible. The 1, 2 or 1, 6 is known as the ortho (o–), the 1, 3 or 1, 5 as meta (m–) and the 1, 4 as para (p–) disubstituted compounds. A few examples of derivatives of benzene are given below:
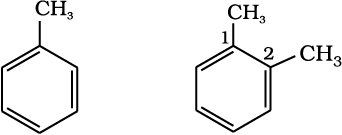
Methylbenzene(Toluene) 1,2-Dimethylbenzene (o-Xylene)
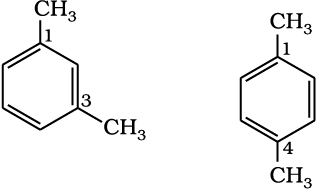
1,3 Dimethylbenzene (m-Xylene) 1,4-Dimethylbenzene ( p-Xylene)

© 2026 GoodEd Technologies Pvt. Ltd.