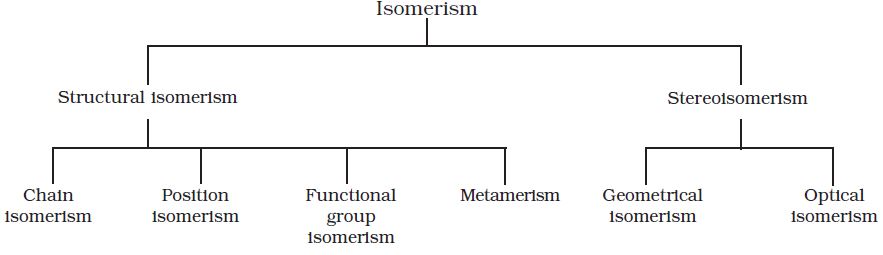
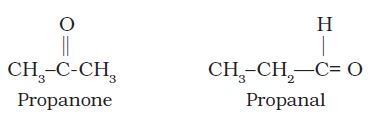The phenomenon of existence of two or more compounds possessing the same molecular formula but different properties is known as isomerism. Such compounds are called as isomers. The following flow chart shows different types of isomerism.
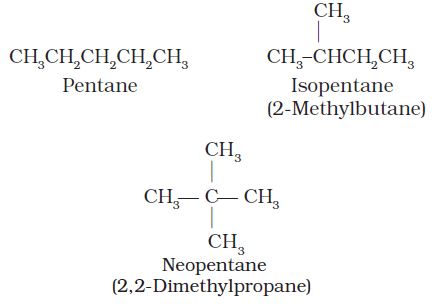
(i) Chain isomerism: When two or more compounds have similar molecular formula but different carbon skeletons, these are referred to as chain isomers and the phenomenon is termed as chain isomerism. For example, C5H12 represents three compounds:
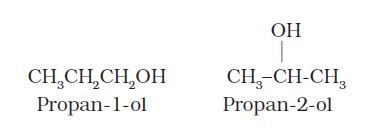
(ii) Position isomerism: When two or more compounds differ in the position of substituent atom or functional group on the carbon skeleton, they are called position isomers and this phenomenon is termed as position isomerism. For example, the molecular formula C3H8O represents two alcohols:
(iii) Functional group isomerism: Two or more compounds having the same molecular formula but different functional groups are called functional isomers and this phenomenon is termed as functional group isomerism. For example, the molecular formula C3H6O represents an aldehyde and a ketone:
(iv) Metamerism: It arises due to different alkyl chains on either side of the functional group in the molecule. For example, C4H10O represents methoxypropane (CH3OC3H7) and ethoxyethane (C2H5OC2H5).

© 2026 GoodEd Technologies Pvt. Ltd.