Orthoboric acid, H3BO3 is a white crystalline solid, with soapy touch. It is sparingly soluble in water but highly soluble in hot water. It can be prepared by acidifying an aqueous solution of borax.
Na2B4O7 + 2HCl + 5H2O → 2NaCl + 4B(OH)3
It is also formed by the hydrolysis (reaction with water or dilute acid) of most boron compounds (halides, hydrides, etc.). It has a layer structure in which planar BO3 units are joined by hydrogen bonds as shown in Fig. 11.1.
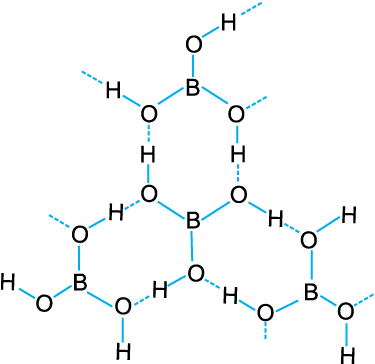
Fig. 11. 1 Structure of boric acid; the dotted lines represent hydrogen bonds
Boric acid is a weak monobasic acid. It is not a protonic acid but acts as a Lewis acid by accepting electrons from a hydroxyl ion:
B(OH)3 + 2HOH → [B(OH)4]– + H3O+
On heating, orthoboric acid above 370K forms metaboric acid, HBO2 which on further heating yields boric oxide, B2O3.
H3BO3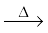 HBO2
HBO2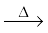 B2O3
B2O3
Problem 11.4
Why is boric acid considered as a weak acid?
Solution
Because it is not able to release H+ ions on its own. It receives OH– ions from water molecule to complete its octet and in turn releases H+ ions.

© 2026 GoodEd Technologies Pvt. Ltd.