Consider a weak acid HX that is partially ionized in the aqueous solution. The equilibrium can be expressed by:

Here, c = initial concentration of the undissociated acid, HX at time, t = 0. α = extent up to which HX is ionized into ions. Using these notations, we can derive the equilibrium constant for the above discussed acid-dissociation equilibrium:
Ka = c2α2 / c(1-α) = cα2 / 1-α
Ka is called the dissociation or ionization constant of acid HX. It can be represented alternatively in terms of molar concentration as follows,
Ka = [H+][X–] / [HX] ( 7.30)
At a given temperature T, Ka is a measure of the strength of the acid HX i.e., larger the value of Ka, the stronger is the acid. Ka is a dimensionless quantity with the understanding that the standard state concentration of all species is 1M.
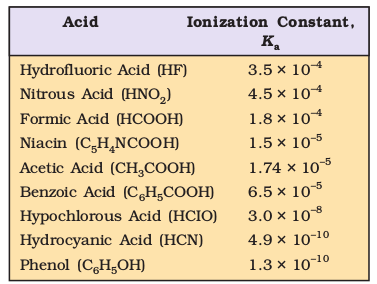
The values of the ionization constants of some selected weak acids are given in Table 7.6.
Table 7.6 The Ionization Constants of Some Selected Weak Acids (at 298K) Acid Ionization Constant
The pH scale for the hydrogen ion concentration has been so useful that besides pKw, it has been extended to other species and quantities. Thus, we have:
pKa = –log (Ka) (7.31)
Knowing the ionization constant, Ka of an acid and its initial concentration, c, it is possible to calculate the equilibrium concentration of all species and also the degree of ionization of the acid and the pH of the solution.
Step 1. The species present before dissociation are identified as Brönsted-Lowry
acids / bases.
Step 2. Balanced equations for all possible reactions i.e., with a species acting both as acid as well as base are written.
Step 3. The reaction with the higher Ka is identified as the primary reaction whilst the other is a subsidiary reaction.
Step 4. Enlist in a tabular form the following values for each of the species in the primary reaction
(a) Initial concentration, c.
(b) Change in concentration on proceeding to equilibrium in terms of α, degree of ionization.
(c) Equilibrium concentration.
Step 5. Substitute equilibrium concentrations into equilibrium constant equation for principal reaction and solve for α.
Step 6. Calculate the concentration of species in principal reaction.
Step 7. Calculate pH = – log[H3O+]
The above mentioned methodology has been elucidated in the following examples.
Problem 7.18
The ionization constant of HF is
3.2 × 10–4. Calculate the degree of dissociation of HF in its 0.02 M solution. Calculate the concentration of all species present (H3O+, F– and HF) in the solution and its pH.
Solution
The following proton transfer reactions are possible:
1) HF + H2O  H3O+ + F–
H3O+ + F–
Ka = 3.2 × 10–4
2) H2O + H2O  H3O+ + OH–
H3O+ + OH–
Kw = 1.0 × 10–14
As Ka >> Kw, [1] is the principle reaction.
HF + H2O  H3O+ + F–
H3O+ + F–
Initial
concentration (M)
0.02 0 0
Change (M)
–0.02α +0.02α +0.02α
Equilibrium
concentration (M)
0.02 – 0.02 α 0.02 α 0.02α
Substituting equilibrium concentrations in the equilibrium reaction for principal reaction gives:
Ka = (0.02α)2 / (0.02 – 0.02α)
= 0.02 α2 / (1 –α) = 3.2 × 10–4
We obtain the following quadratic equation:
α2 + 1.6 × 10–2α – 1.6 × 10–2 = 0
The quadratic equation in α can be solved and the two values of the roots are:
α = + 0.12 and – 0.12
The negative root is not acceptable and hence,
α = 0.12
This means that the degree of ionization, α = 0.12, then equilibrium concentrations of other species viz., HF, F – and H3O+ are given by:
[H3O+] = [F –] = cα = 0.02 × 0.12
= 2.4 × 10–3 M
[HF] = c(1 – α) = 0.02 (1 – 0.12)
= 17.6 × 10-3 M
pH = – log[H+] = –log(2.4 × 10–3) = 2.62
Problem 7.19
The pH of 0.1M monobasic acid is 4.50. Calculate the concentration of species H+, A– and HA at equilibrium. Also, determine the value of Ka and pKa of the monobasic acid.
Solution
pH = – log [H+]
Therefore, [H+] = 10 –pH = 10–4.50
= 3.16 × 10–5
[H+] = [A–] = 3.16 × 10–5
Thus, Ka = [H+][A-] / [HA]
[HA]eqlbm = 0.1 – (3.16 × 10-5) Ä 0.1
Ka = (3.16 × 10–5)2 / 0.1 = 1.0 × 10–8
pKa = – log(10–8) = 8
Alternatively, “Percent dissociation” is another useful method for measure of strength of a weak acid and is given as:
Percent dissociation
= [HA]dissociated/[HA]initial × 100% (7.32)
Problem 7.20
Calculate the pH of 0.08M solution of hypochlorous acid, HOCl. The ionization constant of the acid is 2.5 × 10–5. Determine the percent dissociation of HOCl.
Solution
HOCl(aq) + H2O (l)  H3O+(aq) + ClO–(aq)
H3O+(aq) + ClO–(aq)
Initial concentration (M)
0.08 0 0
Change to reach
equilibrium concentration
(M)
– x + x +x
equilibrium concentartion (M)
0.08 – x x x
Ka = {[H3O+][ClO–] / [HOCl]}
= x2 / (0.08 –x)
As x << 0.08, therefore 0.08 – x Ä 0.08
x2 / 0.08 = 2.5 × 10–5
x2 = 2.0 × 10–6, thus, x = 1.41 × 10–3 [H+] = 1.41 × 10–3 M.
Therefore,
Percent dissociation
= {[HOCl]dissociated / [HOCl]initial }× 100
= 1.41 × 10–3 × 102/ 0.08 = 1.76 %.
pH = –log(1.41 × 10–3) = 2.85.