9.21 Describe the structure of the common form of ice.
The three-dimensional structure of ice is represented as:
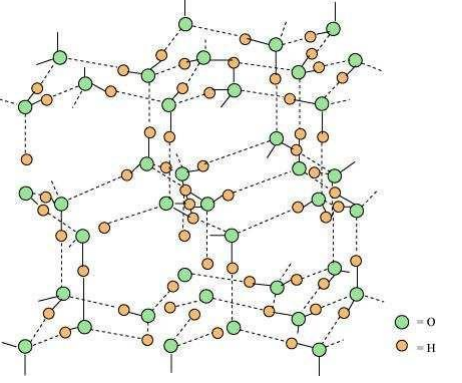
The structure is highly ordered and has hydrogen bonding. Each oxygen atom is surrounded tetrahedrally by four other oxygen atoms at a distance of 276 pm. The structure also contains wide holes that can hold molecules of appropriate sizes interstitially.

© 2026 GoodEd Technologies Pvt. Ltd.