7.20 One of the reaction that takes place in producing steel from iron ore is the reduction of iron(II) oxide by carbon monoxide to give iron metal and CO2.
FeO (s) + CO (g)  Fe (s) + CO2 (g); Kp = 0.265 atm at 1050K
Fe (s) + CO2 (g); Kp = 0.265 atm at 1050K
What are the equilibrium partial pressures of CO and CO2 at 1050 K if the initial partial pressures are: pCO= 1.4 atm and 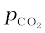 = 0.80 atm?
= 0.80 atm?

© 2026 GoodEd Technologies Pvt. Ltd.