Given below are two statements:
In the light of the above statements, choose the correct answer from the option given below :
1. Both Statement I and Statement II are false.
2. Statement I is correct but Statement II is false.
3. Statement I is incorrect but Statement II is true.
4. Both Statement I and Statement II are true.
| Statement I: | Aniline does not undergo Friedel-Crafts alkylation reaction. |
| Statement II: | Aniline cannot be prepared through Gabriel synthesis. |
1. Both Statement I and Statement II are false.
2. Statement I is correct but Statement II is false.
3. Statement I is incorrect but Statement II is true.
4. Both Statement I and Statement II are true.
Subtopic: Amines - Preparation & Properties | Identification of Primary, Secondary & Tertiary Amines |
64%
From NCERT
NEET - 2024
Please attempt this question first.
Hints
Please attempt this question first.
Match List-I with List-II
Choose the correct answer from the options given below:
1. A-II, B-III, C-I, D-IV
2. A-III, B-II, C-I, D-IV
3. A-II, B-I, C-III, D-IV
4. A-I, B-III, C-II, D-IV
| List-I (Amine) |
List-II \(\textbf{(pK}_{\beta} ~\textbf{value}) \) |
||
| A. | Benzenamine | I. | 3.38 |
| B. | Phenylmethanamine | II. | 9.38 |
| C. | Methenamine | III. | 4.70 |
| D. | N-Ethylethanamine | IV. | 3.0 |
1. A-II, B-III, C-I, D-IV
2. A-III, B-II, C-I, D-IV
3. A-II, B-I, C-III, D-IV
4. A-I, B-III, C-II, D-IV
Subtopic: Identification of Primary, Secondary & Tertiary Amines |
64%
From NCERT
NEET - 2024
Please attempt this question first.
Hints
Please attempt this question first.
Match List-I with List-II
Choose the correct answer from the options given below:
1. A-IV, B-III, C-II, D-I
2. A-IV, B-II, C-III, D-I
3. A-IV, B-II, C-I, D-III
4. A-II, B-III, C-I, D-IV
| List-I (Reagent) |
List-II (name of the reaction) |
||
| A. | Carbylamine test | I. | Phenol |
| B. | Bayer's test | II. | Acetone |
| C. | Iodoform test | III. | Ethylene |
| D. | Phthalein dye test | IV. | Aniline |
Choose the correct answer from the options given below:
1. A-IV, B-III, C-II, D-I
2. A-IV, B-II, C-III, D-I
3. A-IV, B-II, C-I, D-III
4. A-II, B-III, C-I, D-IV
Subtopic: Identification of Primary, Secondary & Tertiary Amines |
78%
From NCERT
NEET - 2024
Please attempt this question first.
Hints
Please attempt this question first.
Identify the compound that will react with Hinsberg's reagent to give a solid which dissolves in alkali.
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Identification of Primary, Secondary & Tertiary Amines |
79%
From NCERT
NEET - 2021
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Which amine gives the carbylamine test?
| 1. | 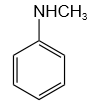 |
2. |  |
| 3. |  |
4. |  |
Subtopic: Identification of Primary, Secondary & Tertiary Amines |
84%
From NCERT
NEET - 2020
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Which amine reacts with Hinsberg's reagent to produce an alkali insoluble product?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Identification of Primary, Secondary & Tertiary Amines |
76%
From NCERT
NEET - 2019
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Mark the correct reaction among the following to convert acetamide to methenamine:
1. Hoffmann bromamide reaction
2. Stephens reaction
3. Gabriels phthalimide synthesis
4. Carbylamine reaction
Subtopic: Amines - Preparation & Properties | Identification of Primary, Secondary & Tertiary Amines |
80%
From NCERT
NEET - 2017
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
An organic compound () (A), when treated with nitrous acid, gave an alcohol and N2 gas was evolved. (A) on warming with CHCl3 and caustic potash gave (C) which on reduction gave isopropylmethylamine. The structure of (A) is:
| 1. | 2. |  |
|
| 3. |  |
4. |  |
Subtopic: Identification of Primary, Secondary & Tertiary Amines |
60%
From NCERT
AIPMT - 2012
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Which compound, upon reduction with lithium aluminium hydride, yields a secondary amine?
1. Nitroethane
2. Methylisocyanide
3. Acetamide
4. Methyl cyanide
Subtopic: Identification of Primary, Secondary & Tertiary Amines | Cyanides & Isocyanides |
81%
From NCERT
AIPMT - 2007
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch


