The chiral centre is absent in:
1. DCH2-CH2-CH2-Cl
2. CH3-CHD-CH2-Cl
3. CH3-CHCl-CH2D
4. CH3-CHOH-CH2-CH3
Which one of the following compounds is most acidic:
| 1. |  |
2. |  |
| 3. |  |
4. | \(\mathrm{Cl}-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{OH}\) |
IUPAC name of some compounds is given. The incorrect name is:
| 1. |  : 3-Methyl-4-ethylheptane : 3-Methyl-4-ethylheptane |
| 2. |  : 3-Methyl-2-butanol : 3-Methyl-2-butanol |
| 3. |  : 2-Ethyl-3-methylbut-1-ene : 2-Ethyl-3-methylbut-1-ene |
| 4. | \(\mathrm{CH}_3-\mathrm{C} \equiv \mathrm{C}-\mathrm{CH}\left(\mathrm{CH}_3\right)_2 \): 4-Methyl-2-pentyne |
The structure of trans-2-hexenal among the following is:
| 1. | 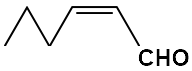 |
| 2. |  |
| 3. |  |
| 4. | None of the above |
The chiral compound among the following is:
| 1. | 2-Methylpentanoic acid | 2. | Pentanoic acid |
| 3. | 4-Methyl pentanoic acid | 4. | None of the above |
The number of chiral carbons in β–D–(+)– glucose is:
1. Six
2. Three
3. Four
4. Five
The compound that is least reactive towards nucleophilic substitution reaction is:
1.
2.
3.
4.
R and S enantiomers differ in:
1. Chemical properties
2. Solubility in achiral solvent
3. Rotation of plane polarised light.
4. Dipole moment
The IUPAC name of the following is:
CH2 = CH – CH2 – CH2 – C ≡ CH
| 1. | 1, 5-Hexenyne | 2. | Hex-1-en-5-yne. |
| 3. | 1-Hexyne-5-ene | 4. | 1, 5-Hexynene |


1. Resonating structures
2. Tautomers
3. Geometrical isomers
4. Optical isomers


