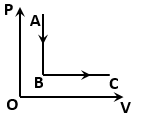
How does the temperature change when the state of an ideal gas is changed according to the process shown in the figure?

1.
temperature increases continuously.
2.
temperature decreases continuously.
3.
temperature first increases and then decreases.
4.
temperature first decreases and then increases.

In the PV graph shown below for an ideal diatomic gas, the change in the internal energy is:
1.
2.
3.
4.
The figure shows a process for a gas in which pressure (P) and volume (V) of the gas change. If and are the molar heat capacities of the gas during the processes AB and BC respectively, then:
1.
2.
3.
4.
The change in the internal energy of an ideal gas does not depend on?
| 1. | Number of moles |
| 2. | Change in temperature |
| 3. | Specific heat at constant pressure \(C_p\) of the gas |
| 4. | Specific heat at constant volume \(C_v\) of the gas |
When the gas in an open container is heated, the mean free path:
| 1. | Increases |
| 2. | Decreases |
| 3. | Remains the same |
| 4. | Any of the above depending on the molar mass |
The translational kinetic energy of oxygen molecules at room temperature is 60 J. Their rotational kinetic energy will be?
1. 40 J
2. 60 J
3. 50 J
4. 20 J
The translational kinetic energy of n moles of a diatomic gas at absolute temperature T is given by:
1.
2.
3.
4.
If the pressure of a gas is doubled, then the average kinetic energy per unit volume of the gas will be:
| 1. | half of its initial value. | 2. | double its initial value. |
| 3. | one-fourth of its initial value. | 4. | four times its initial value. |
When a large bubble rises from the bottom of a lake to the surface, its radius doubles. The atmospheric pressure is equal to that of a column of water of height H. The depth of the lake is:
1. H
2. 2H
3. 7H
4. 8H
How does the pressure of an ideal gas change during the process shown in the diagram?
| 1. | pressure increases continuously. |
| 2. | pressure decreases continuously. |
| 3. | pressure first increases and then decreases. |
| 4. | pressure first decreases and then increases. |