If \(V_\text{H}\),\(V_\text{N}\) and \(V_\text{O}\) denote the root-mean square velocities of molecules of hydrogen, nitrogen and oxygen respectively at a given temperature, then:
1. \(V_\text{N}>V_\text{O}>V_\text{H}\)
2. \(V_\text{H}>V_\text{N}>V_\text{O}\)
3. \(V_\text{O}>V_\text{N}>V_\text{H}\)
4. \(V_\text{O}>V_\text{H}>V_\text{N}\)
Molecular weight of two gases are \(M_1\) and \(M_2.\) At any temperature, the ratio of root mean square velocities \(v_1\) and \(v_2\) will be:
1. \(\sqrt{\frac{M_1}{M_2}}\)
2. \(\sqrt{\frac{M_2}{M_1}}\)
3. \(\sqrt{\frac{M_1+M_2}{M_1-M_2}}\)
4. \(\sqrt{\frac{M_1-M_2}{M_1+M_2}}\)
The root mean square velocity of the molecules of a gas is 300 m/s. What will be the root mean square speed of the molecules if the atomic weight is doubled and absolute temperature is halved?
| 1. | 300 m/s | 2. | 150 m/s |
| 3. | 600 m/s | 4. | 75 m/s |
The molecules of a given mass of gas have rms velocity of 200 ms-1 at \(27^{\circ}\mathrm{C}\) and 1.0 x 105 Nm-2 pressure. When the temperature and pressure of the gas are increased to, respectively, \(127^{\circ}\mathrm{C}\) and 0.05 X 105 Nm-2, rms velocity of its molecules in ms-1 will become:
1. 400/√3
2. 100√2/3
3. 100/3
4.100√2
At which temperature the velocity of \(\mathrm{O_2}\) molecules will be equal to the velocity of \(\mathrm{N_2}\) molecules at \(0^\circ \mathrm{C}?\)
| 1. | \(40^\circ \mathrm{C}\) | 2. | \(93^\circ \mathrm{C}\) |
| 3. | \(39^\circ \mathrm{C}\) | 4. | Cannot be calculated |
The curve between absolute temperature and \(\mathrm{v}^2_{rms}\) is:
| 1. |  |
2. |  |
| 3. |  |
4. | 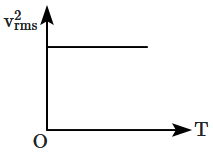 |
The rms speed of oxygen atoms is v. If the temperature is halved and the oxygen atoms combine to form oxygen molecules, then the rms speed will be:
1.
2.
3. 2v
4.
At what temperature is the root mean square speed of molecules of hydrogen twice as that at STP?
1. \(273\) K
2. \(546\) K
3. \(819\) K
4. \(1092\) K
The average translational kinetic energy of \(O_2\) (molar mass \(32\)) molecules at a particular temperature is \(0.048~\text{eV}\). The translational kinetic energy of \(N_2\) (molar mass \(28\)) molecules in \(\text{eV}\) at the same temperature is:
1. \(0.0015\)
2. \(0.003\)
3. \(0.048\)
4. \(0.768\)


