Given below are two statements:
| Assertion (A): | As a gas bubble rises from the bottom of a lake, its volume decreases. |
| Reason (R): | As the gas bubble rises from the bottom of a lake, the pressure of the gas within decreases. |
| 1. | (A) is True but (R) is False. |
| 2. | (A) is False but (R) is True. |
| 3. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
Subtopic: Ideal Gas Equation |
69%
From NCERT
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
The figure shows a plot of \(\frac{PV}{T}\) versus \(P\) for \(1.00 \times 10^{- 3}~\text{kg}\) of oxygen gas at two different temperatures.

The value of \(\frac{PV}{T}\) where the curves meet on the \(y\text-\)axis is:
1. \(0.06~\text{JK}^{-1}\)
2. \(0.36~\text{JK}^{-1}\)
3. \(0.16~\text{JK}^{-1}\)
4. \(0.26~\text{JK}^{-1}\)
Subtopic: Ideal Gas Equation |
58%
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Given below are two statements:
| Assertion (A): | A gas can be liquified at any temperature by an increase of pressure alone. |
| Reason (R): | On increasing pressure, the temperature of gas decreases. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Subtopic: Ideal Gas Equation |
65%
From NCERT
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
For the process \(A\rightarrow B\) of an ideal gas of fixed mass, the density of the gas:
| 1. | increases. |
| 2. | decreases. |
| 3. | remains constant. |
| 4. | first increases then decreases. |
Subtopic: Ideal Gas Equation |
70%
From NCERT
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
PV versus T graph of equal masses of H2, He, and O2 is shown in the figure. Choose the correct alternative:
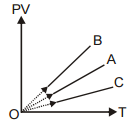
1. A corresponds to H2, B to He, and C to O2
2. A corresponds to He, B to H2, and C to O2
3. A corresponds to He, B to O2, and C to H2
4. A corresponds to O2, B to H2, and C to He
Subtopic: Ideal Gas Equation |
72%
From NCERT
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital



