The change in the internal energy of an ideal gas does not depend on?
1.
Number of moles
2.
Change in temperature
3.
Specific heat at constant pressure \(C_p\) of the gas
4.
Specific heat at constant volume \(C_v\) of the gas
When the gas in an open container is heated, the mean free path:
1. Increases
2. Decreases
3. Remains the same
4. Any of the above depending on the molar mass
The translational kinetic energy of oxygen molecules at room temperature is \(60~\text J.\) Their rotational kinetic energy will be?
1. \(40~\text J\)
2. \(60~\text J\)
3. \(50~\text J\)
4. \(20~\text J\)
Two isotherms are drawn at temperatures as shown. The ratio of mean speed at is:
| 1. | 1: 4 | 2. | 1: 2 |
| 3. | 2: 1 | 4. | 4: 1 |
The translational kinetic energy of \(n\) moles of a diatomic gas at absolute temperature \(T\) is given by:
1. \(\frac{5}{2}nRT\)
2. \(\frac{3}{2}nRT\)
3. \(5nRT\)
4. \(\frac{7}{2}nRT\)
If the pressure of a gas is doubled, then the average kinetic energy per unit volume of the gas will be:
| 1. | half of its initial value. | 2. | double its initial value. |
| 3. | one-fourth of its initial value. | 4. | four times its initial value. |
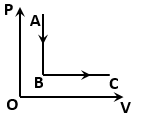
The figure shows a process for a gas in which pressure (P) and volume (V) of the gas change. If and are the molar heat capacities of the gas during the processes AB and BC respectively, then:
1.
2.
3.
4.
In the PV graph shown below for an ideal diatomic gas, the change in the internal energy is:
1.
2.
3.
4.
How does the pressure of an ideal gas change during the process shown in the diagram?
| 1. | pressure increases continuously. |
| 2. | pressure decreases continuously. |
| 3. | pressure first increases and then decreases. |
| 4. | pressure first decreases and then increases. |
The pressure in a diatomic gas increases from to , when its volume is increased from . The increase in internal energy will be:
1.
2.
3.
4.