Consider the following reaction:

The product 'A' is:
1.
2.
3.
4.

Match the compounds given in List-I with List-II and select the suitable option from the code given below:
| List-I | List-II |
|
(a) Benzaldehyde |
(i) Phenolphthalein |
|
(b) Phthalic anhydride |
(ii) Benzoin condensation |
|
(c) Phenyl benzoate |
(iii) Oil of wintergreen |
|
(d) Methyl salicylate |
(iv) Fries rearrangement |
Codes:
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (i) | (iv) | (iii) |
| 2. | (iv) | (i) | (iii) | (ii) |
| 3. | (iv) | (ii) | (iii) | (i) |
| 4. | (ii) | (iii) | (iv) | (i) |
A yellow precipitate with iodine and alkali is given by:
(i). Methyl acetate
(ii). Acetamide
(iii). 2-Hydroxypropane
(iv). Acetophenone
1. (i), and (ii)
2. (ii), and (iii)
3. (iii), and (iv)
4. (iv), and (i)
The compound that does not give an iodoform test is:
1. 3-Pentanone
2. 2-Pentanone
3. Ethanol
4. Ethanal
Ethylbenzene is obtained from phenyl methyl ketone by using:
1. Zn–Hg+HCl
2. LiAlH4
3. KMnO4
4. None of the above
An acid that forms an anhydride (X) on heating and an acid imide (Y) on strong heating with ammonia is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
An organic compound with the molecular formula C9H10O forms a 2,4-DNP derivative, reduces Tollens’ reagent, and undergoes the Cannizzaro reaction. On vigorous oxidation, it gives 1,2-benzenedicarboxylic acid. This organic compound will be:
1. 2-Methyl benzaldehyde
2. 2-Hydroxy benzaldehyde
3. 2-Ethyl benzaldehyde
4. 4-Ethyl benzaldehyde
The below structure is an example of :
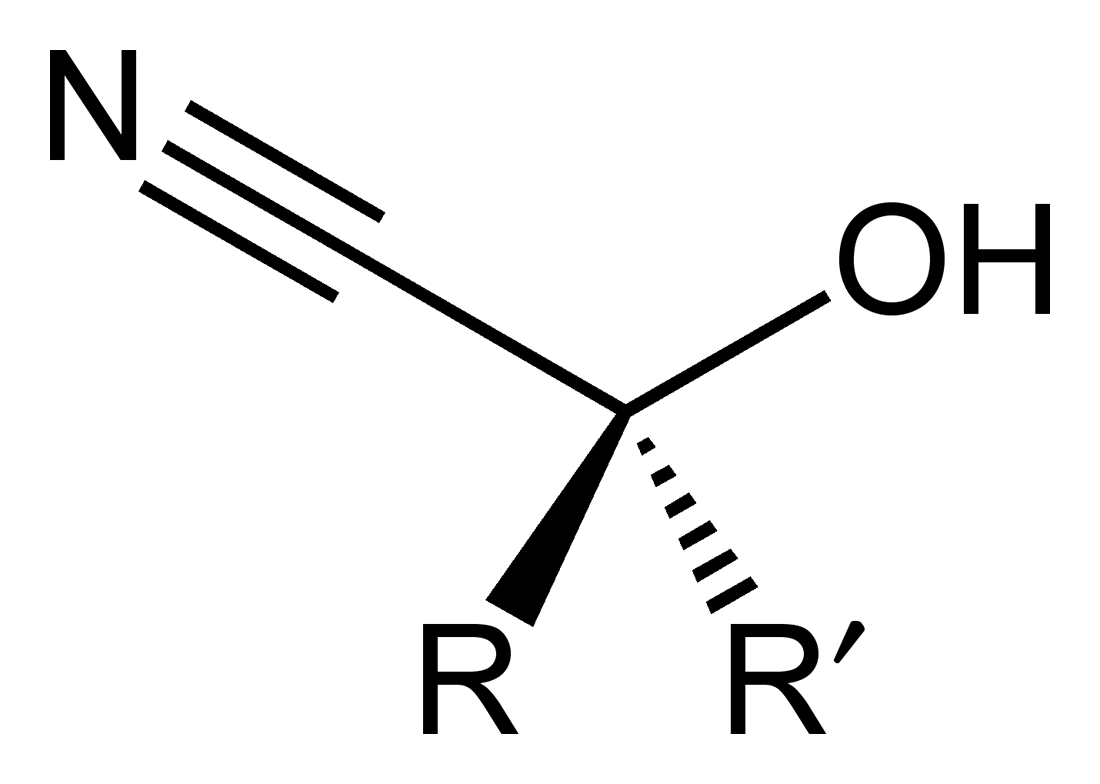
| 1. | Cyanohydrin | 2. | Hemiacetal |
| 3. | Acetal | 4. | Cyanoalcohol |
The IUPAC name for \(\mathrm{CH}_3 \mathrm{COCH}_2 \mathrm{COCH}_3\) is:
1. Pentane-2,4-dione
2. Pentane-1,4-dione
3. Pentane-2,2-dione
4. Pentane-3,4-dione
The structure of Hex-2-en-4-ynoic acid is
| 1. |  |
2. |  |
| 3. |  |
4. | None of these |


