The reaction of toluene with a halogen in the presence of iron (III) chloride gives ortho and para halocompounds,
This can be classified as:
1. Electrophilic elimination reaction
2. Electrophilic substitution reaction
3. Free radical addition reaction
4. Nucleophilic substitution reaction
This can be classified as:
vic-Dihalide among the following is
1. Dichloromethane
2. 1,2-Dichloroethane
3. Ethylidene chloride
4. Allyl chloride
What results from treating chloromethane with excess ammonia?
1. N, N-dimethylmethanamine (\(N{(CH_3)}_{3}\))
2. N-methylmethanamine (CH3-NH—CH3)
3. Methanamine (CH3NH2)
4. Mixture containing all these in equal proportion
The reaction below is an example of :
1. Addition reaction.
2.
3. Elimination reaction.
4.
Which of the following is an enantiomer of the given molecule?

| 1. |  |
2. |  |
| 3. |  |
4. |  |
Match the items in Column I and Column II.
| Column l | Column ll |
| A. SN1 reaction | 1. vic-dibromide |
| B. Elimination of HX from an alkyl halide | 2. Chlorobromocarbons |
| C. Bromination of alkenes | 3. Racemisation |
| D. Chemicals in fire extinguisher | 4. Saytzeff rule |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 3 | 4 | 1 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 1 | 3 | 2 |
Match the structures of compounds given in Column I with the classes of compounds given in Column II.
| Column I | Column II | ||
| A. |  |
1. | Aryl halide |
| B. | CH2=CH-CH2-X | 2. | Alkyl halide |
| C. |  |
3. | Vinyl halide |
| D. | CH2=CH-X | 4. | Allylic halide |
Codes
| A | B | C | D | |
| 1. | 2 | 4 | 1 | 3 |
| 2. | 3 | 4 | 1 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 1 | 3 | 2 |
Match the reactions given in Column I with the types of reactions given in Column II and mark the appropriate option.
| Column I | Column II | ||
| A. |  |
1. | Nucleophilic aromatic substitution reaction |
| B. | 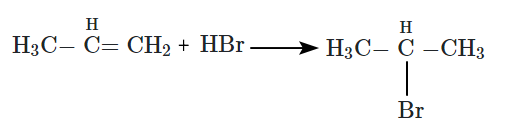 |
2. | Electrophilic aromatic substitution |
| C. |  |
3. | Saytzeff elimination |
| D |  |
4. | Electrophilic addition |
| 5. | Nucleophilic substitution reaction |
Codes
| A | B | C | D | |
| 1. | 2 | 4 | 5 | 1 |
| 2. | 3 | 1 | 5 | 2 |
| 3. | 5 | 4 | 3 | 2 |
| 4. | 4 | 5 | 3 | 2 |
Match the structures given in Column I with the names in Column II.
| Column I | Column II | ||
| A. |  |
1. | 4-Bromopent-2-ene |
| B. |  |
2. | 4-Bromo-3-methylpent-2-ene |
| C. |  |
3. | 1-Bromobut-2-ene |
| D. |  |
4. | 1-Bromo-2-methylpent-2-ene |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 1 | 2 | 3 | 4 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 1 | 3 | 2 |
The correct match of column I (Reactions) with column II (Name of reactions) is:
| Column I | Column II | ||
| A. |  |
1. | Fittig reaction |
| B. |  |
2. | Wurtz-Fittig reaction |
| C. |  |
3. | Finkelstein reaction |
| D. | \(\begin{aligned}\small{C_{2}H_{5}Cl \ + \ NaI \ \xrightarrow[]{\textbf{dry acetone}} \\ \ C_{2}H_{5}I \ + \ NaCl} \end{aligned}\) | 4. | Sandmeyer reaction |
Codes
| A | B | C | D | |
| 1. | 2 | 1 | 4 | 3 |
| 2. | 3 | 1 | 4 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 1 | 3 | 2 |


