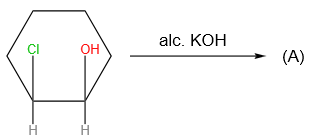
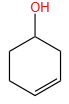
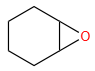
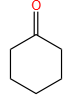
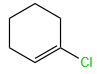
The X and Y in the below mentioned reaction are
| X | Y | |
| 1. | Dilute aqueous NaOH, 20 oC | HBr/Acetic acid, 20 oC |
| 2. | Concentrated alcoholic NaOH, 80 oC | HBr/Acetic acid, 20 oC |
| 3. | Diluted aqueous NaOH, 20 oC, | Br2 / CHCl3 , 0 oC |
| 4. | Concentrated alcoholic NaOH, 80 ° C | Br2/CHCl3, 0 oC |
What is the total number of chiral products produced by monochlorination of 2-methylbutane ?
1. 2
2. 4
3. 6
4. 8
When vinyl chloride is passed through an alcoholic KOH solution, which of the following is correct?
1. It gets dissolved.
2. It forms vinyl alcohol
3. It forms acetylene
4. It has no action
The product (C) in the below-mentioned reaction is:
\(CH_{3}CH_{2}CH_{2}Br \ \xrightarrow[]{\textbf{KOH(alc.)}} \)
\(\ (A) \ \xrightarrow[]{\textbf{HBr}} \ (B) \ \xrightarrow[]{\textbf{KOH(aq}} \ (C)\)
| 1. | Propene | 2. | Propyne |
| 3. | Propan-1-ol | 4. | Propan-2-ol |
\(HC\equiv CNa \ + \ Cl-CH_{2}-CH_{2}-CH_{2}-I \ \rightarrow \ (A) \)
The major product (A) in the above reaction is :
1. H-CC-CH2-CH2-CH2-I
2. CH2=CH-CH2-I
3. H-CC-CH2-CH2-CH2-Cl
4. CH2=CH-CH2-Cl
Which of the following alkyl halides has the maximum density?
| 1. | \(\mathrm{C}_3 \mathrm{H}_7 \mathrm{I} \) | 2. | \(\mathrm{C}_2 \mathrm{H}_5 \mathrm{I} \) |
| 3. | \(\mathrm{CH}_3 \mathrm{I} \) | 4. | \(\mathrm{CH}_3 \mathrm{Br}\) |
| 1. | They show H-bonding. | 2. | They are soluble in water. |
| 3. | They are soluble in organic solvents. | 4. | They do not contain any polar bond. |
The set of reagents used to produce freon (CCl2F2) are:
| 1. | C + F2 + Cl2 → |
| 2. | CH3Cl + F2 → |
| 3. | \(CCl_4 + HF \ \xrightarrow[]{SbCl_5} \) |
| 4. | CCl4 + F2 → |