The number of Faradays required to produce 20.0 g of Ca from molten CaCl2 is-
1. 2F
2. 1F
3. 4F
4. 3F
Three electrolytic cells A, B, C containing solutions of ZnSO4, AgNO3, and CuSO4, respectively are connected in series.
A steady current of 1.5 amperes was passed through them until 1.45 g of silver was deposited at the cathode of cell B. The current flow time is-
1. 14 minutes
2. 25 minutes
3. 20 minutes
4. 11 minutes
The cell in which the following reactions occurs:
has \(E_{cell}^{o}\) = 0.236 V at 298 K.
The equilibrium constant of the cell reaction is :
The cell that will measure the standard electrode potential of a copper electrode is:
| 1. | Pt(s) | H2(g, \(1 \over 10\) bar) | H+(aq, 1M) || Cu2+(aq, 1M) | Cu |
| 2. | Pt(s) | H2(g, 1 bar) | H+(aq, 1M) || Cu2+ (aq, 2M) | Cu |
| 3. | Pt(s) | H2(g, 1 bar) | H+(aq, 1M)|| Cu2+ (aq, 1M) | Cu |
| 4. | Pt(s) | H2(g, \(1 \over 10\) bar) | H+(aq, \(1 \over 10\)M) || Cu2+(aq, 1M) | Cu |
The electrode potential for Mg electrode varies according to the equation
\(E_{Mg^{2+}/Mg}\ = \ E_{Mg^{2+}/Mg}^{o} \ - \ \frac{0.059}{2}log\frac{1}{[Mg^{2+}]}\)
The graph of EMg2+ / Mg vs log [Mg2+] among the following is:
| 1. | 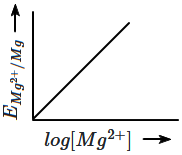 |
2. |  |
| 3. |  |
4. |  |
The correct statement among the following options is:
| 1. | Ecell and ∆rG of cell reaction are both extensive properties. |
| 2. | Ecell and ∆rG of cell reaction are both intensive properties. |
| 3. | Ecell is an intensive property while ∆rG of cell reaction is an extensive property. |
| 4. | Ecell is an extensive property while ∆rG of cell reaction is an intensive property. |
The difference between the electrode potentials of two electrodes when no current is drawn through the cell is called:
| 1. | Cell potential. | 2. | Cell emf. |
| 3. | Potential difference. | 4. | Cell voltage. |
The incorrect statement about an inert electrode in a cell is:
| 1. | It does not participate in the cell reaction. |
| 2. | It provides a surface either for oxidation or for the reduction reaction. |
| 3. | It provides a surface for the conduction of electrons. |
| 4. | It provides a surface for redox reaction. |
An electrochemical cell can behave like an electrolytic cell when -
1. Ecell = 0
2. Ecell > Eext
3. Eext > Ecell
4. Ecell = Eext
The incorrect statement about the solution of electrolytes is:
| 1. | Conductivity of solution depends upon the size of ions. |
| 2. | Conductivity depends upon the viscosity of solution. |
| 3. | Conductivity does not depend upon the solvation of ions present in solution. |
| 4. | Conductivity of solution increases with temperature. |


