Free radical formation will take place in :
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
The arrangement in decreasing order of stability of H3, H5,(CH3)2H and (CH3)3 free radicals is-
1. H3 > H5 > (CH3)2H > (CH3)3
2. (CH3)3 > (CH3)2H > H5 > H3
3. H5 > H3 > (CH3)2H > (CH3)3
4. (CH3)3 > (CH3)2H > H3 >H5
The most stable carbocation among the following is-
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The most stable carbocation among the following is:
| 1. |  |
2. |  |
| 3. | 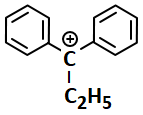 |
4. |  |
A tertiary butyl carbocation is more stable than a secondary butyl carbocation because-
1. + R effect of groups
2. R effect of CH3 groups
3. Hyperconjugation
4. l effect of CH3 groups
The correct statement among the following is -
1. Allyl carbocation (CH2=CH-CH2+) is more stable than propyl carbocation
2. Propyl carbocation is more stable than allyl carbocation
3. Both are equally stable
4. None of the above
The species that contains only three pairs of electrons among the following is-
| 1. | Carbocation | 2. | Carbanion |
| 3. | Free radical | 4. | None of the above |
A paramagnetic species among the following is-
| 1. | A carbocation | 2. | A free radical |
| 3. | A carbanion ion | 4. | All of the above |
The decreasing order of the stability of the given ions is-
| (I). |  |
(II). |  |
| (III). |  |
| 1. | I>II>III | 2. | II>III>I |
| 3. | III>I>II | 4. | II>I>III |
The compound that gives the most stable carbonium ion after C- Cl bond ionisation among the following is-
| 1. |  |
2. |  |
| 3. |  |
4. |  |


