The correct reactivity order towards hydrolysis is:
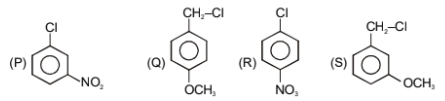
1.
Q>R>S>P
2.
Q>P>R>S
3.
S>R>Q>P
4.
Q>S>R>P

The effect that makes 2,3–dimethyl-2-butene more stable than 2-butene is-
1. Resonance
2. Hyperconjugation
3. Steric effect
4. Inductive effect
Compounds with as molecular formula can exhibit-
1. Position isomerism
2. Metamerism
3. Functional isomerism
4. All of the above

The IUPAC name of the above mentioned compound is -
1. Cyclohexylidenemethanone
2. Cyclohexylidemethanone
3. Cyclohexylidenylmethanone
4. Cyclohexdenemethanone
A tertiary butyl carbocation is more stable than a secondary butyl carbocation because-
1. + R effect of groups
2. R effect of CH3 groups
3. Hyperconjugation
4. l effect of CH3 groups
1. 2–Bromo-3–methylbutanoic acid
2. 2-Methyl-3-bromobutanoic acid
3. 3-Bromo-2-methylbutanoic acid
4. 3-Bromo-2,3-dimethylpropanoic acid.
The pair of structures that does not represent isomers is:
| 1. | 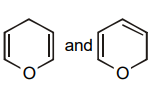 |
2. | 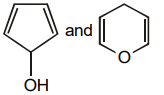 |
| 3. | 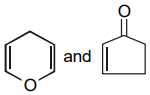 |
4. | 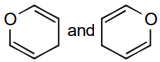 |
The most stable carbocation among the following is-
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The aromatic compound among the following is:-
| 1. | 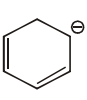 |
2. |  |
| 3. | 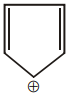 |
4. | 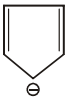 |
The pair among the following that does not contain position isomers is -
| 1. | 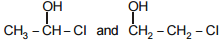 |
| 2. | 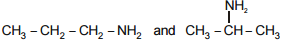 |
| 3. |  |
| 4. |  |



