Combination of atoms A and B that forms an anti-bonding molecular orbital is :
1.
2.
3.
4.
A pair in which both species are not likely to exist is:
| 1. | \(H^+_2,He^{2-}_2\) | 2. | \(H^-_2,He^{2+}_2\) |
| 3. | \(H^{2+}_2,He_2\) | 4. | \(H^-_2,He^{2+}_2\) |
The number of (i) sp2 hybridized carbon atoms and (ii) bonds are present in the following compound are:
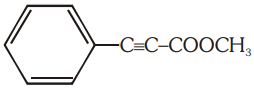
1. 7, 5
2. 8, 6
3. 7, 6
4. 8, 5
Among the following atomic orbital overlaps, the non-bonding overlap is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The decreasing order of ionic character of the N-H, F-H, C-H, and O-H, is :
| 1. | N-H > F-H > C-H > O-H |
| 2. | F-H > N-H > C-H > O-H |
| 3. | O-H > C-H > F-H > N-H |
| 4. | F-H > O-H > N-H > C-H |
The right order of increase in the ionic character of the molecules,
LiF, K2O, N2, SO2, ClF3 is :
1. N2 < SO2 < ClF3 < K2O < LiF
2. N2 > SO2 > ClF3 > K2O < LiF
3. N2 > SO2 > K2O > ClF3 > LiF
4. LiF > K2O < ClF3 > SO2 > N2
From the perspective of molecular orbital theory, which statement is false?
1. is not a stable molecule.
2. is not stable but is expected to exist.
3. Bond strength of is maximum amongst the homonuclear diatomic molecules belonging to the second period.
4. The order of energies of molecular orbitals in molecule is:
The number of sigma () and pi () bonds in pent-2-en-4-yne is:
1. 13 bonds and no bond
2. 10 bonds and 3 bonds
3. 8 bonds and 5 bonds
4. 11 bonds and 2 bonds
Which of the following is the most basic oxide?
1.
2.
3.
4.
An electron-deficient compound among the following is:
| 1. | (SiH3)2 | 2. | (BH3)2 |
| 3. | PH3 | 4. | (CH3)2 |


