The incorrect statement regarding the characteristics of X-rays is :
1. The radiation can ionize the gas
2. It causes a fluorescence effect on ZnS
3. It is deflected by electric and magnetic fields
4. Have a shorter wavelength than ultraviolet rays
For any H-like system, the ratio of velocities of I, II & III orbit i.e., will be:
1. 1 : 2 : 3
2. 1 : 1/2 : 1/3
3. 3 : 2 : 1
4. 1 : 1 : 1
In photoelectric effect, the kinetic energy of photoelectrons increases linearly with the:
1. Wavelength of incident light.
2. Frequency of incident light.
3. Velocity of incident light.
4. Atomic mass of an element.
The photoelectric emission from a surface starts only when the light incident upon the surface has a certain minimum:
| 1. | Intensity | 2. | Wavelength |
| 3. | Frequency | 4. | Velocity |
The ratio of slopes of curves in photoelectric effect gives -
(v=frequency, =maximum kinetic energy, stopping potential):
1. Charge of electron
2. Planck's constant
3. Work function
4. The ratio of Planck's constant of electronic charge
The largest de Broglie wavelength among the following (all have equal velocity) is:
1. molecule
2. molecule
3. Electron
4. Proton
The graphs that represents the variation of momentum of particle with de-Broglie wavelength is:
| 1. |  |
2. | 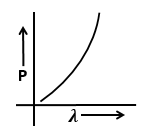 |
| 3. | 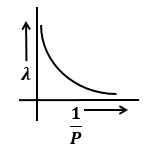 |
4. | 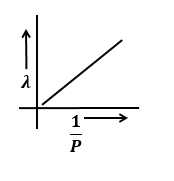 |
The excited state of an H atom emits a photon of wavelength and returns to the ground state. The principal quantum number of the excited state is given by:
1.
2.
3.
4.
A dye absorbs a photon of wavelength and re-emits the same energy into two photons of wavelengths and respectively. The wavelength is related to and as:
1.
2.
3.
4.
The quantum number not obtained from Schrodinger’s wave equation is:
| 1. | \(n\) | 2. | \(l\) |
| 3. | \(m\) | 4. | \(s\) |


