Two rods of the same length and the same area of the cross-section are joined. The temperature of the two ends is shown in the figure.

As we move along the rod, temperature varies as shown in the following figure.

Then:
1. \(K_{1}>K_{2}\)
2. \(K_{1}=K_{2}\)
3. \(K_{1}<K_{2}\)
4. none of these


Then:
1. \(K_{1}>K_{2}\)
2. \(K_{1}=K_{2}\)
3. \(K_{1}<K_{2}\)
4. none of these

| 1. | \(C_{L_1}>C_{L_2}~\text{and }U_1<U_2\) |
| 2. | \(C_{L_2}>C_{L_1}~\text{and }U_1<U_2\) |
| 3. | \(C_{L_1}<C_{L_2}~\text{and }U_1>U_2\) |
| 4. | \(C_{L_1}<C_{L_2}~\text{and }U_1<U_2\) |
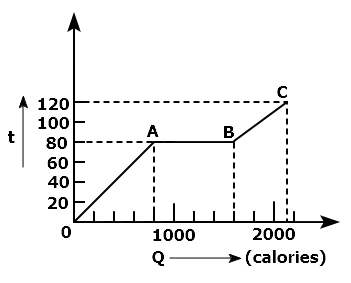
A substance is in solid form at \(0^{\circ}\mathrm{C}\). The amount of heat added to this substance and its temperature are plotted in the following graph. If the relative specific heat capacity of the solid substance is 0.5, from the graph, the specific latent heat of the melting process is: (Specific heat capacity of water = 1000 cal kg-1 K-1 )
| 1. | 60000 cal kg-1 | 2. | 40000 cal kg-1 |
| 3. | 10000 cal kg-1 | 4. | 20000 cal kg-1 |
Liquid oxygen at \(50~\text K\) is heated up to \(300~\text K\) at a constant pressure of \(1~\text{atm}.\) The rate of heating is constant. Which one of the following graphs represents the variation of temperature with time?
| 1. |  |
2. | 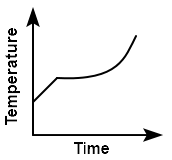 |
| 3. |  |
4. |  |
A body cools in a surrounding which is at a constant temperature of . Assuming that it obeys Newton's law of cooling, its temperature is plotted against time t. Tangents are drawn to the curve at the points A(=1) and B(=2). These tangents meet the time-axis at angles 1 and 2 as shown in the graph then:
1.
2.
3.
4.
In the adjoining figure, various isothermals are shown for a real gas. Then:
| 1. | \(EF\) represents liquification |
| 2. | \(CB\) represents liquification |
| 3. | \(HI\) represents the critical temperature |
| 4. | \(AB\) represents gas at a high temperature |
Radius of a conductor increases uniformly from left end to right end as shown in the figure.
The material of the conductor is isotropic and its curved surface is thermally isolated from its surrounding. Its ends are maintained at temperatures \(T_1\) and \(T_2~(T_1>T_2).\) If in steady state, heat flow rate is equal to \(H,\) then which of the following graphs is correct?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
For a small temperature difference between the body and the surroundings, the relation between the rate of loss heat \(R\) and the temperature of the body \(\theta\) is depicted by:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The adjoining diagram shows the spectral energy density distribution \(E_{\lambda}\) of a black body at two different temperatures. If the areas under the curves are in the ratio \(16:1,\) the value of temperature \(T\) is:

1. \(32000~\text K\)
2. \(16000~\text K\)
3. \(8000~\text K\)
4. \(4000~\text K\)
If \(\lambda_m\) is the wavelength, corresponding to which the radiant intensity of a block is at its maximum and its absolute temperature is \(T,\) then which of the following graphs correctly represents the variation of \(T?\)
| 1. |  |
2. |  |
| 3. |  |
4. |  |