Two identical bodies are made of a material for which the heat capacity increases with temperature. One of these is at \(100~^{\circ}\text{C},\) while the other one is at \(0~^{\circ}\text{C}.\) If the two bodies are brought into contact, then assuming no heat loss, the final common temperature is:
| 1. | \(50~^{\circ}\text{C}\) |
| 2. | more than \(50~^{\circ}\text{C}\) |
| 3. | less than \(50~^{\circ}\text{C}\) but greater than \(0~^{\circ}\text{C}\) |
| 4. | \(0~^{\circ}\text{C}\) |
[Latent heat of ice is \(3.4 \times 10^5~\text{J/kg}\) and \(g=10~\text{N/kg}\)]
1. \(544~\text{km}\)
2. \(136~\text{km}\)
3. \(68~\text{km}\)
4. \(34~\text{km}\)
Steam at \(100~^{\circ}\text{C}\) is passed into \(20~\text{g}\) of water at \(10~^{\circ}\text{C}.\) When water acquires a temperature of \(80~^{\circ}\text{C},\) the mass of water present will be:
[Take specific heat of water \(= 1~\text{cal g}^{-1}~^\circ\text{C}^{-1}\) and latent heat of steam \(= 540~\text{cal g}^{-1}\)]
1. \(24~\text{g}\)
2. \(31.5~\text{g}\)
3. \(42.5~\text{g}\)
4. \(22.5~\text{g}\)
Liquid oxygen at \(50~\text K\) is heated up to \(300~\text K\) at a constant pressure of \(1~\text{atm}.\) The rate of heating is constant. Which one of the following graphs represents the variation of temperature with time?
| 1. |  |
2. | 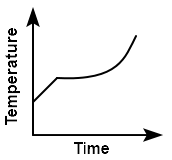 |
| 3. |  |
4. |  |


