Liquid oxygen at \(50~\text K\) is heated up to \(300~\text K\) at a constant pressure of \(1~\text{atm}.\) The rate of heating is constant. Which one of the following graphs represents the variation of temperature with time?
| 1. |  |
2. | 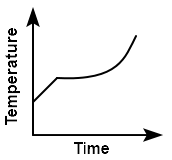 |
| 3. |  |
4. |  |
A piece of ice falls from a height \(h\) so that it melts completely. Only one-quarter of the heat produced is absorbed by the ice, and all energy of ice gets converted into heat during its fall. The value of \(h\) is: (Latent heat of ice is \(3.4\times10^5\) J/kg and \(g=10\) N/kg)
| 1. | \(544\) km | 2. | \(136\) km |
| 3. | \(68\) km | 4. | \(34\) km |
One kilogram of ice at \(0^\circ \text{C}\) is mixed with one kilogram of water at \(80^\circ \text{C}.\) The final temperature of the mixture will be: (Take: Specific heat of water = \(4200~\text{J kg}^{-1}\text{K}^{-1},\) latent heat of ice\(=336~\text{kJ kg}^{-1}\))
| 1. | \(0^\circ \text{C}\) | 2. | \(50^\circ \text{C}\) |
| 3. | \(40^\circ \text{C}\) | 4. | \(60^\circ \text{C}\) |
\(5 ~\text g\) of water at \(30^{\circ} \text{C}\) and \(5 ~\text g\) of ice at \(-20^{\circ} \text{C}\) are mixed together in a calorimeter. The water equivalent of the calorimeter is negligible, and the specific heat and latent heat of ice are \(0.5~\text{cal/g}^{\circ} \text{C}\) and \(80~\text{cal/g},\) respectively. The final temperature of the mixture is:
| 1. | \(0^{\circ} \text{C}\) | 2. | \(-8^{\circ} \text{C}\) |
| 3. | \(-4^{\circ} \text{C}\) | 4. | \(2^{\circ} \text{C}\) |
\(150\) g of ice at \(0^\circ \mathrm{C}\) is mixed with \(100\) g of water at a temperature of \(80^\circ \mathrm{C}.\) The latent heat of ice is \(80\) cal/g and the specific heat of water is \(1\) cal/g°C. Assuming no heat loss to the environment, the amount of ice that does not melt is:
| 1. | \(100\) g | 2. | \(0\) |
| 3. | \(150\) g | 4. | \(50\) g |
Steam at \(100^{\circ}\mathrm{C}\) is injected into 20 g of \(10^{\circ}\mathrm{C}\) water. When water acquires a temperature of \(80^{\circ}\mathrm{C}\), the mass of water present will be: (Take specific heat of water =1 cal g-1 \(^\circ\)C-1 and latent heat of steam = 540 cal g-1)
| 1. | 24 g | 2. | 31.5g |
| 3. | 42.5 g | 4. | 22.5 g |
When the pressure on the surface of water is increased, its boiling point will:
| 1. | Decrease | 2. | Increase |
| 3. | Remain same | 4. | Increase or decrease |
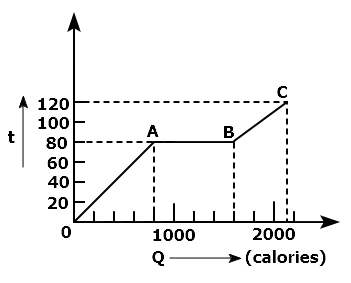
A substance is in solid form at \(0^{\circ}\mathrm{C}\). The amount of heat added to this substance and its temperature are plotted in the following graph. If the relative specific heat capacity of the solid substance is 0.5, from the graph, the specific latent heat of the melting process is: (Specific heat capacity of water = 1000 cal kg-1 K-1 )
| 1. | 60000 cal kg-1 | 2. | 40000 cal kg-1 |
| 3. | 10000 cal kg-1 | 4. | 20000 cal kg-1 |
Some water at \(0^{\circ}\mathrm{C}\) is placed in a large insulated enclosure (vessel). The water vapour formed is pumped out continuously. What fraction of the water will eventually freeze, if the latent heat of vaporization is seven times the latent heat of fusion?
| 1. | 7/8 | 2. | 8/7 |
| 3. | 3/8 | 4. | 5/8 |
Two identical bodies are made of a material whose heat capacity increases with temperature. One of these is at \(100^{\circ} \mathrm{C}\), while the other one is at \(0^{\circ} \mathrm{C}\). If the two bodies are brought into contact, then assuming no heat loss, the final common temperature will be:
| 1. | \(50^{\circ} \mathrm{C}\) |
| 2. | more than \(50^{\circ} \mathrm{C}\) |
| 3. | less than \(50^{\circ} \mathrm{C}\) but greater than \(0^{\circ} \mathrm{C}\) |
| 4. | \(0^{\circ} \mathrm{C}\) |