Steam at \(100~^{\circ}\text{C}\) is passed into \(20~\text{g}\) of water at \(10~^{\circ}\text{C}.\) When water acquires a temperature of \(80~^{\circ}\text{C},\) the mass of water present will be:
[Take specific heat of water \(= 1~\text{cal g}^{-1}~^\circ\text{C}^{-1}\) and latent heat of steam \(= 540~\text{cal g}^{-1}\)]
1. \(24~\text{g}\)
2. \(31.5~\text{g}\)
3. \(42.5~\text{g}\)
4. \(22.5~\text{g}\)
Subtopic: Calorimetry |
72%
From NCERT
AIPMT - 2014
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Liquid oxygen at \(50~\text K\) is heated up to \(300~\text K\) at a constant pressure of \(1~\text{atm}.\) The rate of heating is constant. Which one of the following graphs represents the variation of temperature with time?
| 1. |  |
2. | 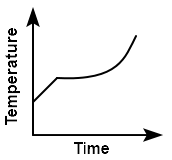 |
| 3. |  |
4. |  |
Subtopic: Calorimetry |
82%
From NCERT
AIPMT - 2012
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
If the cold junction of a thermocouple is kept at \(0^{\circ}\text{C}\) and the hot junction is kept at \(T^{\circ}\text{C}\), then the relation between neutral temperature \((T_{n})\) and temperature of inversion \((T_{i})\) is:
1. \(T_n = \frac{T_i}{2}\)
2. \(T_n = 2T_i\)
3. \(T_n = T_i-T\)
4. \(T_n = T_i+T\)
1. \(T_n = \frac{T_i}{2}\)
2. \(T_n = 2T_i\)
3. \(T_n = T_i-T\)
4. \(T_n = T_i+T\)
Subtopic: Calorimetry |
AIPMT - 2007
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links


