Steam at \(100~^{\circ}\text{C}\) is passed into \(20~\text{g}\) of water at \(10~^{\circ}\text{C}.\) When water acquires a temperature of \(80~^{\circ}\text{C},\) the mass of water present will be:
[Take specific heat of water \(= 1~\text{cal g}^{-1}~^\circ\text{C}^{-1}\) and latent heat of steam \(= 540~\text{cal g}^{-1}\)]
1. \(24~\text{g}\)
2. \(31.5~\text{g}\)
3. \(42.5~\text{g}\)
4. \(22.5~\text{g}\)
[Take specific heat of water \(= 1~\text{cal g}^{-1}~^\circ\text{C}^{-1}\) and latent heat of steam \(= 540~\text{cal g}^{-1}\)]
1. \(24~\text{g}\)
2. \(31.5~\text{g}\)
3. \(42.5~\text{g}\)
4. \(22.5~\text{g}\)
A certain quantity of water cools from \(70~^{\circ}\text{C}\) to \(60~^{\circ}\text{C}\) in the first \(5\) minutes and to \(54~^{\circ}\text{C}\) in the next \(5\) minutes. The temperature of the surroundings is:
1. \(45~^{\circ}\text{C}\)
2. \(20~^{\circ}\text{C}\)
3. \(42~^{\circ}\text{C}\)
4. \(10~^{\circ}\text{C}\)
| 1. | Wien’s displacement Law |
| 2. | Kirchhoff’s Law |
| 3. | Newton’s Law of cooling |
| 4. | Stefan’s Law |
If the radius of a star is \(R\) and it acts as a black body, what would be the temperature of the star at which the rate of energy production is \(Q?\)
\(\left(\sigma~ \text{is Stefan-Boltzmann constant}\right)\)
1. \(\dfrac{Q}{4\pi R^2\sigma}\)
2. \(\left(\dfrac{Q}{4\pi R^2\sigma}\right )^{\dfrac{-1}{2}}\)
3. \(\left(\dfrac{4\pi R^2 Q}{\sigma}\right )^{\dfrac{1}{4}}\)
4. \(\left(\dfrac{Q}{4\pi R^2 \sigma}\right)^{\dfrac{1}{4}}\)
Liquid oxygen at \(50~\text K\) is heated up to \(300~\text K\) at a constant pressure of \(1~\text{atm}.\) The rate of heating is constant. Which one of the following graphs represents the variation of temperature with time?
| 1. |  |
2. | 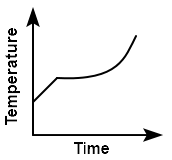 |
| 3. |  |
4. |  |
A cylindrical metallic rod in thermal contact with two reservoirs of heat at its two ends conducts an amount of heat Q in time t. The metallic rod is melted and the material is formed into a rod of half the radius of the original rod. What is the amount of heat conducted by the new rod when placed in thermal contact with the two reservoirs at the same time?
1. Q /4
2. Q/16
3. 2Q
4. Q/2
The total radiant energy per unit area, normal to the direction of incidence, received at a distance \(R\) from the centre of a star of radius \(r,\) whose outer surface radiates as a black body at a temperature \(T\) K is given by: (Where \(\sigma\) is Stefan’s constant):
| 1. | \(\dfrac{\sigma r^{2}T^{4}}{R^{2}}\) | 2. | \(\dfrac{\sigma r^{2}T^{4}}{4 \pi R^{2}}\) |
| 3. | \(\dfrac{\sigma r^{2}T^{4}}{R^{4}}\) | 4. | \(\dfrac{4\pi\sigma r^{2}T^{4}}{R^{2}}\) |
A black body at \(227^{\circ}~\mathrm{C}\) radiates heat at the rate of \(7~ \mathrm{cal-cm^{-2}s^{-1}}\). At a temperature of \(727^{\circ}~\mathrm{C}\), the rate of heat radiated in the same units will be:
1. \(60\)
2. \(50\)
3. \(112\)
4. \(80\)
The two ends of a rod of length L and a uniform cross-sectional area A are kept at two temperatures T1 and T2 (T1> T2). The rate of heat transfer through the rod in a steady state is given by:
1.
2.
3.
4.
On a new scale of temperature, which is linear and called the \(\text{W}\) scale, the freezing and boiling points of water are \(39^\circ ~\text{W}\) and \(239^\circ ~\text{W}\) respectively. What will be the temperature on the new scale corresponding to a temperature of \(39^\circ ~\text{C}\) on the Celsius scale?
1. \(78^\circ ~\text{W}\)
2. \(117^\circ ~\text{W}\)
3. \(200^\circ ~\text{W}\)
4. \(139^\circ ~\text{W}\)




