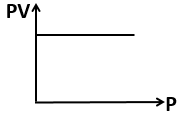
Boyle's law is applicable in :
1. Isobaric process
2. Isochoric process
3. Isothermal process
4. Adiabatic process
At lower temperature, all gases except H2 and He show:
1. Negative deviation
2. Positive deviation
3. Positive and negative deviation
4. None of the above
A gas cannot be liquefied if:
1. Forces of attraction are low under ordinary conditions
2. Forces of attraction are high under ordinary conditions
3. Forces of attraction are zero under ordinary conditions
4. Forces of attraction are either high or low under ordinary conditions
The compressibility factor of an ideal gas is:
1. zero
2. infinite
3. 1
4. -1
The curve that can not represent Boyle's law is -
1.
2.
3.
4.
The deviation of a gas from ideal behavior is expected to be minimum at -
1. 350 K and 3 atm
2. 550 K and 1 atm
3. 250 K and 4 atm
4. 450 K and 2 atm
An ideal gas expands according to PV=constant. On expansion, the temperature of the gas:
1. Will rise.
2. Will drop.
3. Will remain constant.
4. Cannot be determined because the external pressure is not known.
The correct statement for an ideal gas equation is -
1. n is the number of molecules of a gas.
2. V denotes the volume of 1 mole of the gas.
3. n moles of a gas has a volume V.
4. p is the pressure of the gas when only one mole of the gas is present.
The gas among the following that can be most steadily liquefied is -
1. NH3
2. Cl2
3. SO2
4. CO2
The condition of SATP refers to:
1. 25 C and 2 atm
2. 25 C and 1 atm
3. 0 C and 2 atm
4. 25 C and 1 bar