The correct order of the acidic strength for the above compounds is -
 |
 |
 |
| (I) | (II) | (III) |
1. I > II > III
2. III > I > II
3. II > III > I
4. I > III > II

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
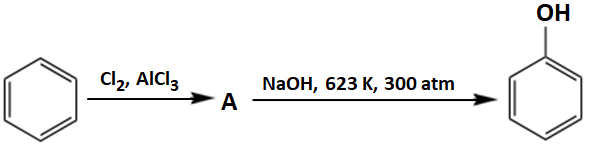
"A" in the reaction below is:
| 1. | Chlorobenzene | 2. | 1,2-Dichlorobenzene |
| 3. | 1,3-dichlorobenzene | 4. | None of these |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The most acidic compound among the following is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Potassium permanganate reagent is used for -
1. Oxidation of primary alcohol to carboxylic acid.
2. Oxidation of primary alcohol to aldehyde.
3. Bromination of phenol to 2,4,6-tribromophenol.
4. Dehydration of propan-2-ol to propene.

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The most acidic compound among the following is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | ||
| A. |  |
1. | Phenetole |
| B. |  |
2. | o-Cresol |
| C. |  |
3. | Catechol |
| D. |  |
4. | Resorcinol |
Codes
| A | B | C | D | |
| 1. | 2 | 4 | 1 | 3 |
| 2. | 3 | 1 | 4 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 3 | 2 | 1 |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Phenol in a set of reactions (as captured below) yielded a product Z.
\(\text{Phenol}\ \xrightarrow{\text{Zn Dust}}\ \text{X}\ \xrightarrow[\text{Anhy. }{AlCl_3}]{CH_3Cl}\ Y\)
\( Y\ \xrightarrow[KMnO_4]{\text{Alkaline}}\ Z\)
The structure of the product Z would be:
1. Toluene
2. Benzaldehyde
3. Benzoic acid
4. Benzene

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The reagent used for the bromination of phenol to 2,4,6-tribromophenol is/are:
1. Bromine water
2. Br2, CS2
3. Br2, CCl4
4. AgBr

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The most acidic compound among the following is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The strongest acid among the following aromatic compounds is -
| 1. | p-Chlorophenol | 2. | p-Nitrophenol |
| 3. | m-Nitrophenol | 4. | o- Nitrophenol |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.