The IUPAC name of the above compound is:

1. 1-Phenylpropan-2-ol
2. 2-Phenylpropan-2-ol
3. 1-Phenylpropan-1-ol
4. 2-Phenylpropan-1-ol

2. 2-Phenylpropan-2-ol
3. 1-Phenylpropan-1-ol
4. 2-Phenylpropan-1-ol

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The IUPAC name of the below compound is:
1. 3,4-Dimethylhexane –1,3,5-triol
2. 3,5-Dimethylhexane –1,1,5-triol
3. 3,5-Dimethylhexane –1,3,5-triol
4. 3,1-Dimethylhexane –1,3,5-triol

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
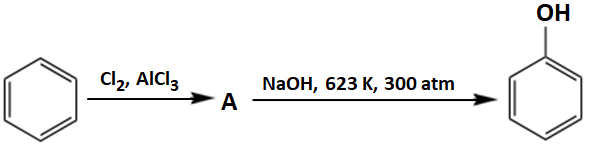
"A" in the reaction below is:
| 1. | Chlorobenzene | 2. | 1,2-Dichlorobenzene |
| 3. | 1,3-dichlorobenzene | 4. | None of these |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Match the following:
| List I | List II | ||
| i. | O2N-C6H5-OCH3 (p) | a. | 4-Ethoxy-1,1-dimethylcyclohexane |
| ii. | C2H5OCH2-CH(CH3)2 | b. | 1-Chloro-2-methoxyethane |
| iii. | CH3OCH2CH2Cl | c. | 1-Ethoxy-2-methylpropane |
| iv. |  |
d. | 1-Methoxy-4-nitrobenzene |
| i | ii | iii | iv | |
| 1. | d | c | b | a |
| 2. | c | d | b | a |
| 3. | a | c | b | d |
| 4. | b | c | d | a |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Preparation of ethers by 2 or 3-degree alcohols in an acidic medium is not a suitable method because:
| 1. | In case of secondary or tertiary alcohols, ketone is obtained as a product. |
| 2. | In case of secondary or tertiary alcohols, aldehyde is obtained as a product. |
| 3. | In case of secondary or tertiary alcohols, alkene is obtained as a product. |
| 4. | In case of primary alcohols, alkene is obtained as a product. |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Propanol has a higher boiling point than that of butane because -
1. Butane undergoes intermolecular ionic bonding
2. Propanol undergoes intermolecular H-bonding
3. Propanol undergoes intramolecular hydrogen bonding
4. Butane undergoes intermolecular H-bonding

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Identify Z in the following reaction:
\(CH_3CH_2CH=CH_2 \xrightarrow[H_2O,\ H_2O_2,\ OH^-]{B_2H_6} Z \)
| 1. | \(CH_3CH_2CH_2CH_2OH\) | 2. | \(CH_3CH_2 \ C \ HCH_3 \\~~~~~~~~~~~~~~~~~~| \\~~~~~~~~~~~~~~~~OH\) |
| 3. | \(CH_3CH_2CH_2CHO \) | 4. | \(CH_3CH_2CH_2CH_3 \) |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The most acidic compound among the following is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Upon dehydration, the below mentioned compound will not give:
| 1. |  |
2. |  |
| 3. |  |
4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
An appropriate set of reactants for the preparation of 1-methoxy-4-nitrobenzene is/are :
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.