Which of the following graph represents the variation of capillary rise of liquid with radius of the capillary tube?
| 1. | 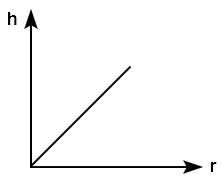 |
2. |  |
| 3. |  |
4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
If the surface tension of water is 0.06 , then the capillary rise in a tube of diameter 1 mm is: \((\theta = 0^{\circ})\)
1. 1.22 cm
2. 2.44 cm
3. 3.12 cm
4. 3.86 cm

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Water rises to height h in a capillary tube. If the length of the capillary tube above the surface of water is made less than h, then:
| 1. | water does not rise at all. |
| 2. | water rises up to the top of the capillary tube, then starts overflowing like a fountain. |
| 3. | water rises up to the top of the capillary tube and stays there without overflowing. |
| 4. | water rises up to a point a little below the top and stays there. |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The wettability of a surface by a liquid depends primarily on:
| 1. | viscosity |
| 2. | surface tension |
| 3. | density |
| 4. | angle of contact between the surface and the liquid |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
If a capillary tube is partially dipped vertically into liquid and the levels of the liquid inside and outside are same, then the angle of contact is:
| 1. | 90° | 2. | 30° |
| 3. | 45° | 4. | 0° |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Water rises to a height \(\mathrm{h}\) in a capillary at the surface of earth. On the surface of the moon, the height of water column in the same capillary will be:
1. \(\mathrm{6h}\)
2.
3. \(\mathrm{h}\)
4. \(\mathrm{zero}\)

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
A liquid filled in a container has plane meniscus. If is the angle of contact, then:
1. = \(0^\circ\)
2. = \(90^\circ\)
3. < \(90^\circ\)
4. = \(180^\circ\)

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
If the capillary experiment is performed in a vacuum, then for a liquid the capillary will:
1. rise
2. remain the same
3. fall
4. rise to the top

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Three liquids of densities (with ), having the same value of surface tension T, rise to the same height in three identical capillaries. The angles of contact obey:
| 1. | \(\frac{\pi}{2}>\theta_1>\theta_2>\theta_3 \geq 0\) |
| 2. | \(0 \leq \theta_1<\theta_2<\theta_3<\frac{\pi}{2}\) |
| 3. | \(\frac{\pi}{2}<\theta_1<\theta_2<\theta_3<\pi\) |
| 4. | \(\pi>\theta_1>\theta_2>\theta_3>\frac{\pi}{2}\) |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
A capillary tube of radius \(r\) is immersed in water and water rises in it to a height \(h.\) The mass of the water in the capillary is \(5\) g. Another capillary tube of radius \(2r\) is immersed in water. The mass of water that will rise in this tube is:
1. \(5.0\) g
2. \(10.0\) g
3. \(20.0\) g
4. \(2.5\) g

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.


