Select the correct option based on statements below:
Assertion (A):
Dissolution of sugar in water proceeds via an increase in entropy.
Reason (R):
Entropy decreases, when an egg is boiled hard.
1.
Both (A) and (R) are true and (R) is the correct explanation of (A).
2.
Both (A) and (R) are true but (R) is not the correct explanation of (A).
3.
(A) is true but (R) is false.
4.
Both (A) and (R) are false.

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
| Assertion (A): | Work done in an irreversible isothermal process at constant volume is zero. |
| Reason (R): | Work is assigned a negative sign during expansion and is assigned a positive sign during compression. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
| Assertion (A): | Specific heat is an intensive property. |
| Reason (R): | Heat capacity is an extensive property. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The thermodynamic stability of NO(g) based on the above data is:
1. Less than NO2(g)
2. More than NO2(g)
3. Equal to NO2(g)
4. Insufficient data

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
For the graph given below, it can be concluded that work done during the process shown will be-
| 1. | Zero | 2. | Negative |
| 3. | Positive | 4. | Cannot be determined |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Consider the following diagram for a reaction .
The nature of the reaction is-
1. Exothermic
2. Endothermic
3. Reaction at equilibrium
4. None of the above

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
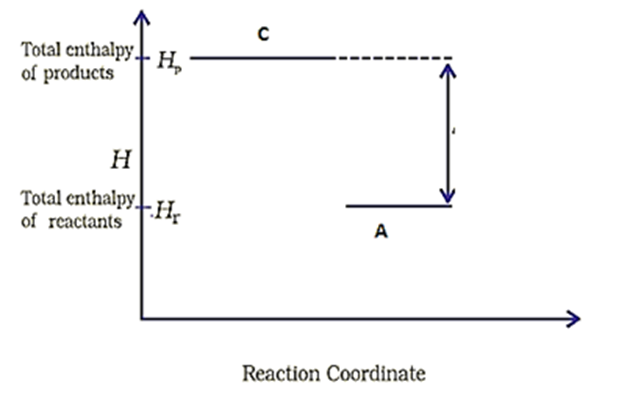
Consider the following diagram for a reaction
The nature of the reaction is-
| 1. | Exothermic | 2. | Endothermic |
| 3. | Reaction at equilibrium | 4. | None of the above |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The equilibrium constant for a reaction is 10. The value of will be:
( )

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The standard enthalpy of the formation of CH3OH(l) from the following data is:
| \(\small{\mathrm{CH}_3 \mathrm{OH}_{(l)}+\frac{3}{2} \mathrm{O}_2(\mathrm{g}) \rightarrow \mathrm{CO}_2(\mathrm{g})+2 \mathrm{H}_2 \mathrm{O}_{(l)} \text {; }}\) \( \Delta_{\mathrm{r}} \mathrm{H}^{\circ}=-726 \mathrm{~kJ} \mathrm{~mol}{ }^{-1}\) |
| \(\small{\mathrm{C}(\mathrm{s})+\mathrm{O}_2(\mathrm{g}) \rightarrow \mathrm{CO}_2(\mathrm{g}) \text {; } }\) \(\Delta_{\mathrm{c}} \mathrm{H}^{\circ}=-393 \mathrm{~kJ} \mathrm{~mol}{ }^{-1}\) |
| \(\small{\mathrm{H}_{2(\mathrm{g})}+\frac{1}{2} \mathrm{O}_{2(\mathrm{g})} \rightarrow \mathrm{H}_2 \mathrm{O}_{(l)} \text {; } } \) \(\Delta_{\mathrm{f}} \mathrm{H}^{\circ}=-286 \mathrm{~kJ} \mathrm{~mol}^{-1}\) |
| 1. | −239 kJ mol−1 | 2. | +239 kJ mol−1 |
| 3. | −47 kJ mol−1 | 4. | +47 kJ mol−1 |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
. The standard enthalpy of formation of gas in the above reaction would be-
| 1. | -92.4 J (mol)-1 | 2. | -46.2 kJ (mol)-1 |
| 3. | +46.2 J (mol)-1 | 4. | +92.4 kJ (mol)-1 |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.