The correct statement regarding the comparison of staggered and eclipsed conformations of ethane is:
1.
The eclipsed conformation of ethane is more stable than staggered conformation because eclipsed conformation has no torsional strain.
2.
The eclipsed conformation of ethane is more stable than staggered conformation even though the eclipsed conformation has a torsional strain.
3.
The staggered conformation of ethane is more stable than eclipsed conformation because staggered conformation has no torsional strain.
4.
The staggered conformation of ethane is less stable than eclipsed conformation because staggered conformation has a torsional strain.

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
X and Y in the above-mentioned reaction are respectively:
1. X = 2–Butyne; Y = 3–Hexyne
2. X = 2-Butyne; Y = 2-Hexyne
3. X = 1-Butyne; Y = 2-Hexyne
4. X = 1-Butyne; Y = 3–Hexyne

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
HCl with an alkene X reacts in accordance with Markovnikov’s rule to give 1-Chloro-1-methylcyclohexane. The structure of alkene (X) is:
| 1. |  |
2. |  |
| 3. | (1) and (2) | 4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The reaction of C6H5CH =CHCH3 with HBr produces:
1. \(C_{6} H_{5} \underset{Br}{\underset{\left|\right.}{CH}} CH_{2} CH_{3} \)
2. \(C_{6} H_{5} CH_{2} \underset{Br}{\underset{\left|\right.}{CH}} CH_{3} \)
3. \(C_{6} H_{5} CH_{2} CH_{2} CH_{2} Br \)
4.

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Above mentioned single molecule is obtained from ozonolysis. The starting cyclic compound is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Given compounds are as follows:
| (I) |  |
(II) | 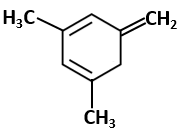 |
| (III) |  |
The enthalpy of hydrogenation of these compounds will be in the order as-
| 1. | I > II > III | 2. | III > II > I |
| 3. | II > III > I | 4. | II > I > III |
Product Z in the above-mentioned reaction is:
1.
2.
3.
4.

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The isobutyl group among the following is:
| 1. | |
| 2. | CH3–CH2–CH2–CH2– |
| 3. | 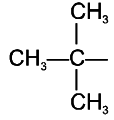 |
| 4. |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The incorrect IUPAC name among the following is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
| 1. |  |
2. |  |
| 3. |  |
4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.







