Compound A () is treated with H2O/H2SO4 gives , an optically inactive compound. The structure of A is-
1.
CH3CH2CH=CH2
2.
CH3CH=CHCH3
3.
(CH3)2C=CH2
4.


Compound A is:
| 1. |  |
2. |  |
| 3. |  |
4. | 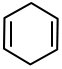 |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The major product in the above-mentioned reaction is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The increasing order of the reactivity of the following compounds towards electrophilic aromatic substitution reaction is:
| 1. | III < I < II | 2. | III < II < I |
| 3. | II < I < III | 4. | II < III < I |
In the following reaction,
\(\mathrm{ CH_{3}C\equiv CH\xrightarrow[Red \ hot \ iron \ tube]{873 K} \ A}\)
The number of () bonds present in the product (A) is:
1. 21
2. 9
3. 24
4. 18

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
3-Phenylpropene on reaction with HBr gives (as a major product)-
1.
2.
3.
4.
1,2-dibromopropane on treatment with X moles of followed by treatment with ethyl bromine gave a pent-2-yne. The value of X is -
1. 1
2. 2
3. 3
4. 4

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.

The product X in the above-mentioned reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The reaction among the following that proceeds through an electrophilic substitution reaction is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The most suitable reagent for the following conversion is-
1. Hg2+/ H+, H2O
2. Na/liquid NH3
3. H2, Pd/C, quinoline
4. Zn/HCl

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.






