In the presence of peroxide, hydrogen chloride and hydrogen fluoride do not undergo anti-Markovnikov's
addition reaction with alkenes because-
1. Both are highly ionic
2. One is oxidizing and the other is reducing
3. One of the steps is endothermic in both the cases
4. All the steps are exothermic in both cases

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The reaction of propene with HOCl proceeds via the addition of-
| 1. | H+ in the first step | 2. | Cl+ in the first step |
| 3. | OH- in the first step | 4. | Cl+ and OH- in a single step |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The compound among the following that yield propyne on hydrolysis is:
1.
2.
3.
4.

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The reaction of HBr with
in the presence of peroxide will give:
| 1. | 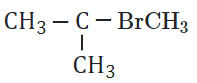 |
| 2. |  |
| 3. |  |
| 4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
An alkene among the following that reacts fastest with H2 under catalytic conditions is -
| 1. |  |
2. |  |
| 3. |  |
4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The product(s) obtained via oxymercuration (HgSO4 + H2SO4 ) of but-1-yne is -
1.
2. CH3CH2 CH2-CHO
3. CH3CH2CHO + HCHO
4. CH3 CH2COOH + HCOOH

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The most acidic compound among the following is-
1.
2.
3.
4.

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Ethene and ethyne can be distinguished by:
1. Br2 water
2. KMnO4 solution
3. Cuprous chloride solution
4. All of the above
The reaction intermediate involved in the addition of \(\mathrm{HBr}\) to propene in the absence of peroxides is:
| 1. | \(H^+\) | 2. | \(Br^-\) |
| 3. | \(H \) | 4. | \(Br\) |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Which of the following Hydrocarbon has the highest octane number?
1. Methane
2. Ethane
3. Iso-octane
4. Triptane



