The incorrect statement among the following for a nucleophile is:
1. Nucleophile is a Lewis acid
2. Ammonia is a nucleophile
3. Nucleophiles attack low electron density sites
4. Nucleophiles are not electron seeking

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
In Duma's method of estimation of nitrogen, 0.35 g of an organic compound gave 55 ml of nitrogen collected at 300 K temperature and 715 mm pressure. The percentage composition of nitrogen in the compound would be:
(Aqueous tension at 300 K = 15 mm)
1. 16.45
2. 27.45
3. 44.45
4. 35.45

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The most acidic compound among the following is-
| 1. |  |
2. |  |
| 3. |  |
4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
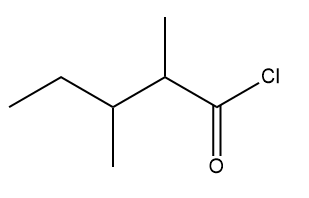
The IUPAC name of the above mentioned compound is -
1. 3, 4-Dimethylpentanoyl chloride
2. 1-Chloro-1-oxo-2,3-dimethylpentane
3. 2-Ethyl-3-methylbutanoylchloride
4. 2, 3-Dimethylpentanoyl chloride

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The enolic form of ethyl acetoacetate is given below. The number of sigma and pi bonds in the enolic form of ethyl acetoacetate are -
1. 18 sigma bonds and 2 pi-bonds
2. 16 sigma bonds and 1 pi-bond
3. 9 sigma bonds and 2 pi-bonds
4. 9 sigma bonds and 1 pi-bond

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The compound that gives the most stable carbonium ion after C- Cl bond ionisation among the following is-
| 1. |  |
2. |  |
| 3. |  |
4. |  |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The correct order of acidity among the following is:
1. CH2=CH2 >CH≡CH > CH3C≡CH > CH3-CH3
2. CH≡CH > CH3-C≡CH > CH2=CH2 >CH3-CH3
3. CH≡CH > CH2=CH2 > CH3-C≡CH > CH3-CH3
4. CH3-CH3 > CH2=CH2 > CH3-C≡CH > CH≡CH

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The optically active compound among the following is-
1. Isobutyric acid
2. beta-Chloropropionic acid
3. Propionic acid
4. alpha-Chloropropionic acid

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Glucose and fructose are-
| 1. | Chain isomers | 2. | Position isomers |
| 3. | Functional isomers | 4. | Optical isomers |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The optically active compound among the following is-
| 1. | Glycerine | 2. | Acetaldehyde |
| 3. | Glyceraldehyde | 4. | Acetone |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.



