If the electronic configuration of an element is , the four electrons that participate in the chemical bond formation will be from :
1.
2.
3.
4.

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The electronic configurations of the elements A, B, and C are given below.
The stable form of A may be represented by the formula:
| 1. | A | 2. | A2 |
| 3. | A3 | 4. | A4 |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The electronic configurations of the elements A, B, and C are given below.
The stable form of C may be represented by the formula :
| 1. | C | 2. | C2 |
| 3. | C3 | 4. | C4 |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The electronic configurations of the elements A, B, and C are given below.
The bond between B and C will be :
| 1. | Ionic. | 2. | Covalent. |
| 3. | Hydrogen. | 4. | Coordinate. |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Most covalent compound among the following is :
| 1. | AlF3 | 2. | AlCl3 |
| 3. | AlBr3 | 4. | AlI3 |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The correct order of covalent bond character among
LiCl, BeCl2, BCl3 and CCl4, is:
1. LiCl < BeCl2 > BCl3 > CCl4
2. LiCl > BeCl2 < BCl3 < CCl4
3. LiCl < BeCl2 < BCl3 < CCl4
4. LiCl > BeCl2 > BCl3 > CCl4

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Among the following, the maximum covalent character is shown by which compound:
1.
2.
3.
4.

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
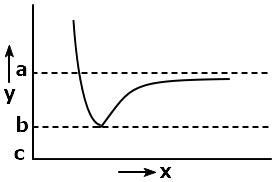
The following graph captures potential energy on the y-axis for hydrogen gas formation as a function of the internuclear distance on the x-axis:
The bond energy of H2 can be represented by-
| 1. | (c – a) | 2. | (b – a) |
| 3. | (c-a)/2 | 4. | (b-a)/2 |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Which of the following atomic orbitals would have the greatest overlap to form the strongest covalent bond?
| 1. | 2s-2s (σ) | 2. | 2s-2p (σ) |
| 3. | 2p-2p (π) | 4. | 2p-2p (σ) |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.