What is the major product when aniline reacts with excess Br2 / H2O?
| 1. | 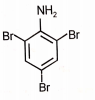 |
2. | 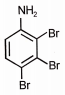 |
| 3. | 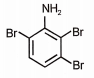 |
4. | 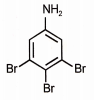 |
1. \(NaOH\)
2. \(HCl\)
3. \(NaOH/Br_2\)
4. \(HgO\)
1. Gabriel phthalimide synthesis.
2. Wurtz's reaction.
3. Hofmann's method.
4. Hinsberg's method.
Match the structures in Column-I with their corresponding names in Column-II:
| Column-I (Structure) | Column-II (Name) | ||
| A. |  |
I. | Benzene-1,2-diamine |
| B. |  |
II. | Propane-1,2-diamine |
| C. |  |
III. | Prop-2-en-1-amine |
| D. |  |
IV. | N, N-Dimethylaniline |
Select the correct option:
1. (A) → (II), (B) → (IV), (C) → (I), (D) → (III)
2. (A) → (I), (B) → (II), (C) → (III), (D) → (IV)
3. (A) → (IV), (B) → (III), (C) → (I), (D) → (II)
4. (A) → (IV), (B) → (III), (C) → (II), (D) → (I)
Reduction of aromatic nitro compounds using Fe and HCl gives:
1. Aromatic oxime
2. Aromatic hydrocarbon
3. Aniline
4. Aromatic amide
| Assertion (A): | Reduction of nitrobenzene using Fe and HCl is preferred over Sn and HCl. |
| Reason (R): | With Fe, only a small amount of HCl is required to initiate the reaction, as FeCl2 formed gets hydrolyzed to release hydrochloric acid. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
The X and A in the following reaction is:
CH3–CH2–Cl + Ethanolic NaCN→ X \(\xrightarrow[]{A}\) CH3–CH2–CH2–NH2
1. X = CH3CH2CN; A = LiAlH4
2. X = CH3CH2NC; A = KMnO4
3. X = CH3CH2CN; A = CrO3
4. X = CH3CH2NC; A = LiAlH4
Which chemical reaction leads to the formation of isocyanide?
1. Reimer Tiemann reaction.
2. Carbylamine reaction.
3. Hoffmann bromamide reaction.
4. None of the above.
Hofmann bromamide degradation reaction is shown by:
| 1 | PhNH2 | 2. | PhCONH2 |
| 3. | PhNO2 | 4. | PhCH2NH2 |
| 1. | Arylamines are generally more basic than alkylamines because the nitrogen lone-pair electrons are not delocalized by interaction with the aromatic ring -electron system. |
| 2. | Arylamines are generally more basic than alkylamines because of aryl group. |
| 3. | Arylamines are generally more basic than alkylamines because the nitrogen atom in arylamines is sp-hybridized. |
| 4. | Arylamines are generally less basic than alkylamines because the nitrogen lone-pair electrons are delocalized by interaction with the aromatic ring -electron system. |






