In which of the following representations given below spatial arrangement of group/atom is different from that given in structure 'A'?

a.

b.

c.

d.

Choose the correct option
1. (a, b, c)
2. (b, c, d)
3. (a,c,d)
4. (a, d, b)





Choose the correct option
Electrophiles are electron-seeking species. Which of the following groups contain only electrophiles?
(a) \(\mathrm {BF_3, NH_3, H_2O} \)
(b) \(\mathrm {AlCl_3, SO_3 , NO_2^+} \)
(c) \(\mathrm {NO_2^+ , CH_3^+, CH_3-C^+\text=O} \)
(d) \(\mathrm {C_2H_5^-,H_3C -\dot{C}H_2,C_2H_5^+} \)
Choose the correct option:
| 1. | (a, b) | 2. | (b, c) |
| 3. | (c, d) | 4. | (a, d) |
Consider the following four compounds:
| (I) |  |
| (II) |  |
| (III) |  |
| (IV) |  |
Which of the following pairs are position isomers?
| 1. | I and II | 2. | II and III |
| 3. | II and IV | 4. | III and IV |
Consider the following four compounds.
| I. | 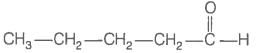 |
| II. | 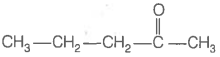 |
| III. | 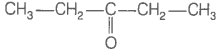 |
| IV. | 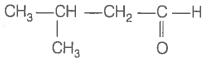 |
Consider the following pairs of compounds.
(a) II and III
(b) II and IV
(c) I and IV
(d) I and II
Choose the pairs given above that are not functional group isomers:
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, c)
A nucleophile is a species that should have:
| (a) | A pair of electrons to donate |
| (b) | Positive charge |
| (c) | Negative charge |
| (d) | Electron-deficient species |
Choose the correct option:
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, c)
| a. | Electrons of carbon-hydrogen \(\sigma \text -\) bond of an alkyl group directly attached to an atom of unsaturated system. |
| b. | Electrons of carbon-hydrogen \(\sigma \text -\) bond of alkyl group directly attached to the positively charged carbon atom. |
| c. | \(\pi \text -\) electrons of carbon-carbon bond. |
| d. | Lone pair of electrons. |
Choose the correct option:
| 1. | (a, b) | 2. | (b, c) |
| 3. | (c, d) | 4. | (a, c) |
Two of the following compounds contain all the carbon atoms in the same hybridisation state.
(a)
(b)
(c)
(d)
Choose the correct option:
1. a and b
2. b and c
3. c and d
4. a and d
Arrange the following carbanions in order of their decreasing stability:
A. \(\mathrm{H}_3 \mathrm{C}-\mathrm{C} \equiv \mathrm{C}^{-}\)
B. \(\mathrm{H}-\mathrm{C} \equiv \mathrm{C}^{-}\)
C. \(\mathrm{H}_3 \mathrm{C}-\mathrm{{C}H_2^-}\)
| 1. | A > B > C | 2. | B > A > C |
| 3. | C > B > A | 4. | C > A > B |
Given below are two statements:
| Assertion (A): | Components of a mixture of red and blue inks can be separated by distributing the components between stationary and mobile phases in paper chromatography. |
| Reason (R): | The colored components of inks migrate at different rates because paper selectively retains different components according to the difference in their partition between the two phases. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
| Assertion (A): | Sulphur present in an organic compound can be estimated quantitatively by the Carius method. |
| Reason (R): | Sulphur is separated easily from other atoms in the molecule and gets precipitated as a light yellow solid. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |




