C and Si both have same lattice structure,having 4 bonding electrons in each.However, C is insulator whereas Si is intrinsic semiconductor. This is because
1. in case of C, the valence band is not completely filled at absolute zero temperature
2. in case of C,the condition band is partly filled even at absolute zero temperature
3. the four bonding electrons in the case of C lie in the second orbit,Whereas in the case of Si they lie in the third
4 .the four bonding electrons in the case of C lie in the third orbit, whereas for Si they lie in the fourth orbit
The figure shows a logic circuit with two inputs A and B and the output C. The voltage wave forms across A, B and C are as given. The logic circuit gate is
1. OR gate
2. NOR gate
3. AND gate
4. NAND gate
The input resistance of a silicon transistor is
100 . Base current is changed by 40
which results in a change in collector current
by 2 mA. This transistor is used as a common-
emitter amplifier with a load resistance of 4 k.
The voltage gain of the amplifier is
1. 2000
2. 3000
3. 4000
4. 1000
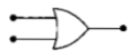
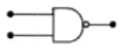
Symbolic representation of four logic gates
are shown as
(i)
(ii)
(iii)
(iv)
Pick out which ones are for AND, NAND and
NOT gates, respectively.
1. (iii), (ii)and (i)
2. (iii), (ii) and (iv)
3. (ii), (iv) and (iii)
4. (ii), (iii) and (iv)
If a small amount of antimony is added to germanium crystal
1. the antimony becomes an acceptor atom
2. there will be more free electrons than holes in the semiconductor
3. its resistance is increased
4. it becomes a p-type semiconductor
In forward biasing of the p-n junction
| 1. | the positive terminal of the battery is connected to n-side and the depletion region becomes thin |
| 2. | the positive terminal of the battery is connected to n-side and the depletion region becomes thick |
| 3. | the positive terminal of the battery is connected to p-side and the depletion region becomes thin |
| 4. | the positive terminal of the battery is connected to p-side and the depletion region becomes thick |
In the following figure, the diodes which are forward biased, are
| (I) |  |
(II) |
 |
| (III) |  |
| (IV) |  |
Choose the correct option from the given ones:
1. III and IV only2. I and III only
3. II only
4. II and IV only
| 1. | \(\mathrm{n}\text-\)type with electron concentration \(n_{e}=5\times10^{22}~\text{m}^{-3}\) |
| 2. | \(\mathrm{p}\text-\)type with electron concentration \(n_{e}=2.5\times10^{23}~\text{m}^{-3}\) |
| 3. | \(\mathrm{n}\text-\)type with electron concentration \(n_{e}=2.5\times10^{10}~\text{m}^{-3}\) |
| 4. | \(\mathrm{p}\text-\)type with electron concentration \(n_{e}=5\times10^{9}~\text{m}^{-3}\) |
A Zener diode, having breakdown voltage equal to \(15\) V, is used in a voltage regulator circuit, as shown in the figure. The current through the diode is:
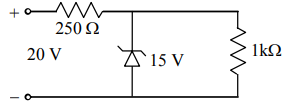
1. \(10\) mA
2. \(15\) mA
3. \(20\) mA
4. \(5\) mA
Which one of the following statement is false?
(1) Pure Si doped with trivalent impurities gives a p-type semiconductor
(2) Majority carries in a n-type semiconductor are holes
(3) Minority carries in a p-type semiconductor are electrons
(4) The resistance of intrinsic semiconductor decreases with increase of temperature