A thermodynamic system is taken through the cycle ABCD as shown in figure. Heat rejected by the gas during the cycle is
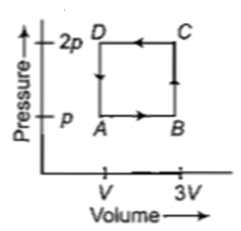
1. 2 pV
2. 4 pV
3.
4. pV

2. 4 pV
3.
4. pV
One mole of an ideal gas from an initial state A undergoes via two processes. It first undergoes isothermal expansion from volume V to 3V and then its volume is reduced from 3V to V at constant pressure. The correct P-V diagram representing the two processes is -
1. 
2. 
3. 
4. 
An ideal gas goes from state \(A\) to state \(B\) via three different processes, as indicated in the \(P\text-V\) diagram. If \(Q_1,Q_2,Q_3\) indicates the heat absorbed by the gas along the three processes and \(\Delta U_1, \Delta U_2, \Delta U_3\) indicates the change in internal energy along the three processes respectively, then:

| 1. | \({Q}_1>{Q}_2>{Q}_3 \) and \(\Delta {U}_1=\Delta {U}_2=\Delta {U}_3\) |
| 2. | \({Q}_3>{Q}_2>{Q}_1\) and \(\Delta {U}_1=\Delta {U}_2=\Delta {U}_3\) |
| 3. | \({Q}_1={Q}_2={Q}_3\) and \(\Delta {U}_1>\Delta {U}_2>\Delta {U}_3\) |
| 4. | \({Q}_3>{Q}_2>{Q}_1\) and \(\Delta {U}_1>\Delta {U}_2>\Delta {U}_3\) |
A mass of diatomic gas (=1.4) at a pressure of 2 atm is compressed adiabatically so that its temperature rise from to The pressure of the gas is final state is-
(1) 28 atm
(2) 68.7 atm
(3) 256 atm
(4) 8 atm
If represent the increase in internal energy and work done by the system respectively in a thermodynamical process,which of the following is true?
(1) in a adiabatic process
(2) in a isothermal process
(3) in adiabatic process
(4) in a isothermal process
A monoatomic gas at pressure and is compressed adiabatically to its original volume. What is the final pressure of the gas ?
1,
2.
3.
4.
The internal energy change in a system that has absorbed 2 kcal of heat and done 500 J of work is
1. 8900 J
2. 6400 J
3. 5400 J
4. 7900 J
In thermodynamic processes, which of the following statements is not true?
| 1. | In an adiabatic process, the system is insulated from the surroundings. |
| 2. | In an isochoric process, the pressure remains constant. |
| 3. | In an isothermal process, the temperature remains constant. |
| 4. | In an adiabatic process, \(P V^\gamma\) = constant. |
If Q, E and W denote respectively the heat added, change in internal energy and the work done in a closed cyclic process, then
1. W=0
2. Q=W=0
3. E=0
4. Q=0
The specific heat at constant pressure and at constant volume for an ideal gas are and and its adiabatic and isothermal elasticities are and respectively. The ratio of to is
1.
2.
3.
4.






