A gas is compressed isothermally to half its initial volume. The same gas is compressed separately through an adiabatic process until its volume is again reduced to half. Then:
1.
compressing the gas through an adiabatic process will require more work to be done.
2.
compressing the gas isothermally or adiabatically will require the same amount of work to be done.
3.
which of the case (whether compression through isothermal or through the adiabatic process) requires more work to be done will depend upon the atomicity of the gas.
4.
compressing the gas isothermally will require more work to be done.
Figure below shows two paths that may be taken by a gas to go from a state \(A\) to a state \(C.\) In process \(AB,\) \(400\text{ J}\) of heat is added to the system and in process \(BC,\) \(100\text{ J}\) of heat is added to the system. The heat absorbed by the system in the process \(AC\) will be-
1. \(380\text{ J}\)
2. \(500\text{ J}\)
3. \(460\text{ J}\)
4. \(300\text{ J}\)
One mole of an ideal diatomic gas undergoes a transition from A to B along a path AB as shown in the figure.
The change in internal energy of the gas during the transition is
1. 20 kJ
2. -20 kJ
3. 20 J
4. -12 kJ
An ideal gas is compressed to half its initial volume by means of several processes.
Which of the following processes results in the maximum work being done on the gas?
1. Adiabatic
2. Isobaric
3. Isochoric
4. Isothermal
The coefficient of performance of a refrigerator is 5. If the temperature inside freezer is -20°C, the temperature of the surroundings to which it rejects heat is -
1. 31°C
2. 41°C
3. 11°C
4. 21°C
A thermodynamic system undergoes cyclic process ABCDA as shown in figure. The work done by the system in the cycle is
1. ρoVo
2. 2ρoVo
3. ρoVo/2
4. zero
A gas is taken through the cycle A→B→C→A, as shown. What is the net work done by the gas?
1. 2000J
2. 1000J
3. Zero
4. -2000J
During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio of CP/CV for the gas is equal to:
| 1. | 4/3 | 2. | 2 |
| 3. | 5/3 | 4. | 3/2 |
A thermodynamic system is taken through the cycle ABCD as shown in figure. Heat rejected by the gas during the cycle is
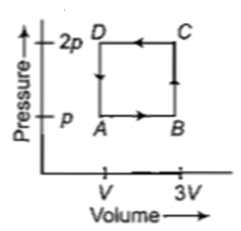
1. 2 pV
2. 4 pV
3.
4. pV
One mole of an ideal gas from an initial state A undergoes via two processes. It first undergoes isothermal expansion from volume V to 3V and then its volume is reduced from 3V to V at constant pressure. The correct P-V diagram representing the two processes is -
1. 
2. 
3. 
4. 










