For a gas, if the ratio of specific heats at constant pressure and constant volume is , then the value of degree of freedom is
(1)
(2)
(3)
(4)
The value \(\gamma = \frac{C_P}{C_V}\) for hydrogen, helium, and another ideal diatomic gas \(X\) (whose molecules are not rigid but have an additional vibrational mode), are respectively equal to:
1. \(\frac{7}{5}, \frac{5}{3}, \frac{9}{7}\)
2. \(\frac{5}{3}, \frac{7}{5}, \frac{9}{7}\)
3. \(\frac{5}{3}, \frac{7}{5}, \frac{7}{5}\)
4. \(\frac{7}{5}, \frac{5}{3}, \frac{7}{5}\)
What is the ratio of temperatures
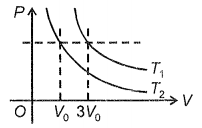
1.
2. 3 : 1
3. 9 : 1
4. 27 : 1
How does the temperature change when the state of an ideal gas is changed according to the process shown in the figure?
| 1. | temperature increases continuously. |
| 2. | temperature decreases continuously. |
| 3. | temperature first increases and then decreases. |
| 4. | temperature first decreases and then increases. |
From the following V-T diagram, we can conclude
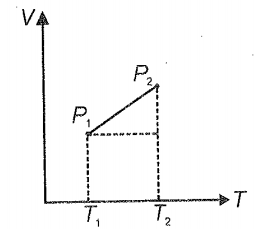
1. =
2. >
3. <
4.
When the gas in an open container is heated, the mean free path:
| 1. | Increases |
| 2. | Decreases |
| 3. | Remains the same |
| 4. | Any of the above depending on the molar mass |
Maxwell's speed distribution graph is drawn as shown below. The most probable speed of the gas molecules is:
1. 4 km/s
2. Between 3 km/s and 1 km/s
3. Any value between 2 km/s and 6 km/s
4. More than 4 km/s
A closed container having an ideal gas is heated gradually to increase the temperature by 20% The mean free path will become/remain:
1. 20% more
2. Same
3. 20% less
4. 33% less
Two ideal gases have the same number of molecules per unit volume and the radii of their molecules are \(r\) and \(3r\) respectively. The ratio of their mean free path in identical containers will be:
1. \(3:1\)
2. \(9:1\)
3. \(1:1\)
4. \(1:4\)
An increase in the temperature of a gas-filled in a container would lead to:
| 1. | decrease in intermolecular distance. |
| 2. | increase in its mass. |
| 3. | increase in its kinetic energy. |
| 4. | decrease in its pressure. |


