You have been provided with three test tubes. One of them contains distilled water and the other two contain an acidic solution and a basic solution, respectively. If you are given only red litmus paper, how will you identify the contents of each test tube?
Why should curd and sour substances not be kept in brass and copper vessels?
Which gas is usually liberated when an acid reacts with a metal? Illustrate with an example. How will you test for the presence of this gas?
Metal compound A reacts with dilute hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle. Write a balanced chemical equation for the reaction, if one of the compounds formed is calcium chloride.
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
Why do HCl, HNO3 etc., show acidic character in aqueous solutions while solutions of compounds like alcohol and glucose do not show acidic character?
Why does an aqueous solution of an acid conduct electricity?
Why does dry HCl gas not change the colour of dry litmus paper?
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
How is the concentration of hydronium ions H3O+ affected when a solution of an acid is diluted?
How is the concentration of hydroxide ions (OH-) affected when excess base is dissolved in a solution of sodium hydroxide?
You have two solutions A and B. The pH of solution A is 6 and pH of solution B is 8.
(i) Which solution is acidic and which is basic?
(ii) Which solution has more H+ ion concentration?
What effect does the concentration of H+(aq) ions have on the nature of the solution?
Do basic solution also have H+ (aq) ions? If yes, then why are these basic?
Under what soil condition do you think a farmer would treat the soil of his fields with quicklime (calcium oxide) or slaked lime (calcium hydroxide) or chalk (calcium carbonate)?
What is the common name of the compound CaOCl2?
Name the substance which on treatment with chlorine yields bleaching powder.
Name the sodium compound which is used for softening hard water.
What will happen if a solution of sodium hydrogen carbonate is heated? Give the equation of the reaction involved.
Write an equation to show the reaction between plaster of Paris and water.
A solution turns red litmus blue, its pH is likely to be
1. 1
2. 4
3. 5
4. 10
A solution reacts with crushed egg-shells to give a gas that turns lime water milky. The solution contains
1. NaCl
2. HCl
3. LiCl
4. KCl
10 mL of a solution of NaOH is found to be completely neutralised by 8mL of a given solution of HCl. If we take 20 mL of the same solution of NaOH, the amount of HCl solution required to neutralise it will be
1. 4 mL
2. 8 mL
3. 12 mL
4. 16 mL
Which one of the following types of medicines is used for treating indigestion?
1. Antibiotic
2. Analgesic
3. Antacid
4. Antiseptic
Write word equations and then balanced equations for the reaction taking place when
(i) dilute sulphuric acid reacts with zinc granules.
(ii) dilute hydrochloric acid reacts with magnesium ribbon.
(iii) dilute sulphuric acid reacts with aluminium powder.
(iv) dilute hydrochloric acid reacts with iron filings.
Compounds such as alcohols and glucose also contain hydrogen but are not categorised as acids. Describe an activity to prove it.
Why does distilled water not conduct electricity, whereas rainwater does?
Why do not acids show acidic behaviour in the absence of water?
Five solutions A, B, C, D and E when tested with universal indicator showed pH as 4, 1, 11, 7 and 9 respectively. Which solution is
(i) neutral (ii) strongly alkaline (iii) strongly acidic (iv) weakly acidic (v) weakly alkaline?
Arrange the pH in increasing order of H+ ion concentration.
Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid (CH3COOH) is added to test tube B. Amount and concentration taken for both the acids are same. In which test tube, will the fizzing occur more vigorously and why?
Fresh milk has a pH of 6. How do you think the pH will change as it turns into curd? Explain.
A milkman adds a very small amount of baking soda to fresh milk.
(i) Why does he shift the pH of the fresh milk from 6 to slightly alkaline?
(ii) What do you expect to observe when milk comes to boil?
Or
What is the effect of the addition of baking soda to milk?
(iii) Why does this milk take a longer time to set as a curd?
Plaster of Paris should be stored in moisture-proof containers. Explain why?
What is a neutralisation reaction? Give two examples.
Give two important uses of washing soda and baking soda.
What is the pH of gastric juice, which is released during digestion?
If a few drops of concentrated acid accidentally spills over the hand of a student, what should be done?
A few drops of liquid X were added to distilled water. It was observed that the pH of the water decresed. What could be the liquid sample X?
A student has four samples A, B, C, D containing dil. HCl, aqueous KCl, dil. NaOH and distilled water respectively. Which two samples would show an equal value of pH?
What happens when nitric acid is added to egg shell?
During the preparation of hydrogen chloride gas on a humid day, the gas is usually passed through the guard tube containing calcium chloride. What is the role of calcium chloride taken in the guard tube?
Why does 1M HCl solution have a higher concentration of H+ ions than 1M CH3COOH solution?
Two solutions A and B have pH values of 5 and 8 respectively. Which solution will be basic in nature?
How does the flow of acid rainwater into a river make the survival of aquatic life in the river difficult?
Arrange the following in increasing order of their pH values: NaOH solution, blood, lemon juice.
Why is acetic acid called a weak acid though there are four H-atoms in the molecule?
How does a string acid differ from a concentrated acid?
Which gas is evolved when sodium hydrogen carbonate (NaHCO3) is added to acetic acid?
Name three chemicals in which common salt are used as the raw material.
Out of two salts baking soda and washing soda, which salt does not contain water of crystallisation?
What happens when a solution of an acid is mixed with a solution of a base in the test tube?
Which gas would be evolved, if sodium bicarbonate is treated with tartaric acid?
Write the name and chemical formula of the products formed by heating gypsum at 373 K.
A solution has pH of 7. Explain how would you
(i) increase its pH (ii) decrease its pH?
Name two crystalline substances which do not contain water of crystallisation.
Write two observations you make when quicklime is added to water.
What is observed when sulphur dioxide is passed through
(i) water? (ii) lime water?
Also write chemical equations for the reactions that the place.
An aqueous solution turns red litmus solution blue. Excess addition of which solution would reverse the change?
State the pH of distilled water and rain water. Justify your answer with proper reasoning.
Choose strong/weak acid and strong/weak bases from the following compounds:
Name a salt of a strong acid like HNO3 and a weak base like NH4OH. Represent the reaction that takes place.
How would you distinguish between baking powder and washing soda by heating?
A calcium compound which is a yellowish white powder is used as a disinfectant and also in the textile industry.
(i) Name the compound.
(ii) Which gas is released when this compound is left exposed to air?
While eating food, you spill some curry on your white shirt. You immediately scrub it with soap. What happens to its yellow colour on scrubbing with soap? Why? What happens to this stain when the shirt is washed with plenty of water?
The oxide of a metal M was water soluble. When a blue litmus strip was dipped in this solution, it did not undergo any change in colour. Predict the nature of the oxide.
Identify the number of replaceable hydrogen ions in the following acids:
(i) (ii)
(ii) (iv)
Replaceable ions.
Farmers are using a large number of pesticides and fertilisers in their fields to increase crop production and to enhance their profits. But by doing so, they are causing damage to the soil as well as to the environment. Do you agree with this statement? Why should we avoid eating fruits and vegetables without washing them properly?
On rubbing a tarnished copper vessel with lemon, it begins to shine again. Explain the reaction.
A substance X is used as a building material and insoluble in water. When reacts with dil. HCl, it produces a gas which turns lime water milky. Predict the substance and write the chemical equation involved.
Why is tartaric acid added into the baking soda to get baking powder?
A compound X of sodium is commonly used in kitchen for making crispy pakoras. It is also used for curing acidity in the stomach. Identify X. What is its chemical formula? State the reaction which takes place when it is heated during cooking.
Why does tooth decay start when the pH of the mouth is lower than 5.5?
Write the chemical formula of washing soda. How is it obtained from baking soda? Name one industrial use of washing soda other than washing clothes.
What is meant by water of crystallisation? How would you show that copper sulphate crystals contain water of crystallisation?
Name the gas which is liberated when an acid reacts with a metal. Illustrate with an example. How will you test the presence of this gas?
Write the chemical formula of bleaching powder. How bleaching powder is prepared? For what purpose it is used in drinking water?
When zinc metal is treated with a dilute solution of a strong acid, a gas is evolved, which is utilised in the hydrogenation of oil.
(i) Name the gas evolved.
(ii) Write the chemical equation of the reaction involved and also write a test to detect the gas formed.
How the following substances will dissociate to produce ions in their solutions?
(i) Hydrochloric acid
(ii) Nitric acid
(iii) Sulphuric acid
(iv) Sodium hydroxide
(v) Potassium hydroxide
(vi) Magnesium hydroxide
Account for the following:
(i) Antacid tablets are used by a person suffering from acidity.
(ii) Toothpaste is used for cleaning teeth.
(iii) Which acid is stinging hair of nettle leaves?
What pH do you expect for the following salt solutions and why?
What is tooth enamel chemically? State the condition when it starts corroding. What happens when food particles left in the mouth after eating degrades? Why do doctors suggest the use of tooth powder/toothpaste to prevent tooth decay?
Or
Tooth enamel is one of the hardest substances in our body. How does it undergo damage due to the eating of chocolates and sweets? What should we do to prevent it?
Baking soda is used in small amount for making bread and cake. It helps to make these soft and spongy. An aqueous solution of baking soda turns red litmus blue. It is also used in soda acid fire extinguisher. Use this information to answer the following questions:
(i) How does it help in extinguishing fire?
(ii) What is the reaction involved when it is heated?
(iii) Is the pH value of baking soda solution lower than or higher than 7?
Answer the following:
(i) Why is sodium hydrogen carbonate an essential ingredient in most antacids?
(ii) When electricity is passed through an aqueous solution of sodium chloride, three products are obtained. Why is the process called
chlor-alkali process?
(i) A chemical compound X is used in glass and soap industry. Identify the compound and give its chemical formula.
(ii) How many molecules of water of crystallisation are present in compound X?
(iii) How will you prepare the above compound starting from sodium chloride? Write all relevant equations involved in the process.
Write the name and formula of each of the following:
(i) An acidic salt
(ii) A basic salt
(iii) A neutral salt
Answer the following:
(i) What happens when crystals of washing soda are left in dry air?
(ii) Name the change that takes place. Which two industries are based on the use of washing soda?
(iii) With the help of balanced chemical equation, state the reaction that takes place when sodium hydrogen carbonate is heated during
cooking.
(i) Explain how anhydrous copper sulphate can be used to detect the presence of moisture in a liquid?
(ii) What is meant by amphoteric oxide?
(iii) Give the chemical name and formula of chloride of lime.
A dry pellet of a common base B when kept in open absorbs moisture and turns sticky. The compound is also a by-product of chor-alkali process.
(i) Identify B.
(ii) What type of reaction occurs when B is treated with an acidic oxide?
(iii) Write a balanced chemical equation for one such solution.
In one of the industrial processes used for the manufacture of sodium hydroxide, a gas X is formed as by-product.
The gas X reacts with lime water to give a compound Y which is used as a bleaching agent in the chemical industry. Identify X and Y by giving the chemical equation of the reactions involved.
Write the chemical name and formula of bleaching powder. How it is prepared? Write the chemical equation and state any two uses of bleaching powder.
(i) Name one natural source of each of the following acids.
(a) Citric acid (b) Oxalic acid
(d) Lactic acid (d) Tartaric acid
(ii) When ions is commonly produced by all acids?
State reason for the following:
(i) Dry HCl gas does not change the colour of the dry blue litmus paper.
(ii) Alcohol and glucose also contain hydrogen, but do not conduct electricity.
(iii) Concentration of ions is affected when a solution of an acid is diluted.
How is Plaster of Paris chemically different from gypsum? How can they be interconverted? Write two uses of Plaster of Paris.
In the following schematic diagram for the preparation of hydrogen gas as shown in the figure, what would happen if the following changes are made?
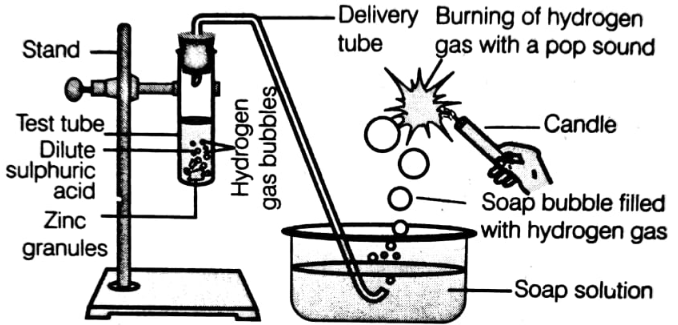
(i) In place of zinc granules, the same amount of zinc dust is taken in the test tube.
(ii) Instead of dilute sulphuric acid, dilute hydrochloric acid is taken.
(iii) In place of zinc, copper turnings are taken.
(iv) Sodium hydroxide is taken in place of dilute sulphuric acid and the test tube is heated.
Give one example in each case:
(i) A basic oxide which is soluble in water.
(ii) A basic oxide which is insoluble in water.
(iii) A weak mineral acid.
(iv) A base which is not an alkali.
(v) A hydrogen containing compound which is not an acid.
What is water of crystallisation? Write the common name and chemical formula of a commercially important compound which has ten water molecules as water of crystallisation. How is the compound obtained? Write the chemical equation also. List any two uses of this compound.
A metal carbonate X on heating with an acid gives a gas which when passed through a solution Y gives the carbonate back. On the other hand, a gas G that is obtained at anode during electrolysis of brine is passed on dry Y, it gives a compound Z, used for disinfecting drinking water. Identify X, Y, G and Z.
(i) Write three physical properties each of acids and bases.
(ii) How will you show with an example that metal oxides are basic in nature? Give the chemical equation also.
(i) Define a universal indicator. Mention its one use.
(ii) Solution A gives pink colour when a drop of phenolphthalein indicator is added to it.
Solution B gives red colour when a drop of methyl orange is added to it.
What type of solutions A and B and which one of the solutions A and B will have higher pH value.
(iii) Name one salt whose solution has pH more than 7 and one salt whose solution has pH less than 7.
(i) Define universal indicator. For what purpose it is used?
(ii) Two solutions A and B have pH values of 3.0 and 9.5 respectively. Which of these will turn litmus solution from blue to red and which
will sum phenolphthalein from colourless to pink.
(iii) Water is a neutral substance. What colour will you get when you add a few drops of universal indicator to a test tube containing
distilled water?
In an attempt to demonstrate electrical conductivity through an electrolyte, the following apparatus was set up.
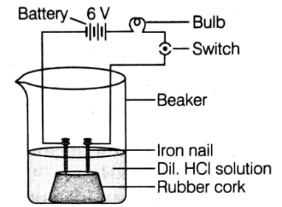
What would you suggest, the bulb will glow in this case or not?
A student dropped few pieces of marble in dil. HCl contained in a test through lime water.
(i) What change would be observed in lime water?
(ii) Write balanced chemical equation for the above change.
A student prepared solutions of an acid and a base in two separate beakers. She forgot to label the solutions and litmus paper is not available in the laboratory. Since, both the solutions are colourless, how will she distinguish between the two?
Brushing our teeth twice a day is a well-known saying. Justify this statement. And what values are associated with it?
A group of students, while on excursion trip is camping on the hills. One morning, they find themselves engulfed in a thick blanket of snow. One of the senior member of the group suggests to sprinkle common salt on the ice slit covering the pavement.
Answer the following questions.
(i) What is the purpose of sprinking common salt on ice slit?
(ii) Can we use any other substance in place of common slat?
(iii) What values are associated with the passage?
Madhu was asked to determine the melting point of a given organic solid. For this, she used a bath containing conc. . When she was looking at the thermometer, she lost her concentration and became a little casual. The beaker containing boiling sulphuric acid fell on her clothes. Her clothes were burnt and she got server burns on hands.
Answer the following questions.
(i) Why did she server burns on her hands?
(ii) What precautions do you suggest which she should take in future?
(iii) What are the values do you infer from this passage?
Rahul was playing with his friends. Suddenly, Rahul was stung by a honeybee and was in great pain. Immediately, his friends call his mother. She applied a coating of toothpaste on the affected area and then took him to the doctor.
Read the above passage and answer the following questions.
(i) What could be the reason for this burning pain?
(ii) Why did his mother applied toothpaste on the affected area?
(iii) What qualities are possessed by Rahul's friends?