Mark the incorrect one with respect to Exotic species
(1) Eupatorium has invaded forests in the northeast replacing Teak
(2) Nile perch a small predator fish introduced in lake Victoria of East America
(3) Water hyacinth was introduced in India for its beautiful flowers and shape of leaves
(4) Lantana camara has replaced native species in many forest of UP and MP
Which of the given is the major cause of species extinction?
(1) Pollution
(2) Coextinction
(3) Habitat loss
(4) Alien species
Select the incorrect match
(1) National forest policy - 1988
(2) Environment (protection)Act - 1986
(3) Air (prevention and control of pollution)Act - 1982
(4) Water (prevention and control of pollution)Act - 1974
Growth in plants is
1. Under the control of external factors only
2. Extrinsic and quantitative phenomenon
3. Manifested by apparent growth only
4. Intrinsic with plasmatic growth also
Global warming is caused by green-house gases and is supposed to be responsible for many possible negative effects, but some positive effects are also there, like
(1) Rising of sea level
(2) Changing the rainfall pattern, helping proper distribution of
(3) Maintaining earth's average temperature at present value
(4) Warming of trophosphere
Mark the correct one with respect to Los Angeles smog
(1) It contains
(2) There is no role of secondary pollutant
(3) Formed at low temperature
(4) Its components can inhibit ETS
Brown air or haze is produced by
(1)
(2)
(3) VOC's
(4) CO
Fundamental elements of taxonomy are
1. Characterisation and classification only
2. Characterisation, identification and classification only
3. Characterisation, classification and nomenclature only
4. Characterisation, identification, classification and nomenclature
which side of amino acid is joined to t-RNA during translation?
1
2 COOH - side
3 H - side
4 R - side
Select the correct sequence of passive absorption path of water in a plant
Select correct match with respect to column I and column II
Column I Column II
a. Albugo (i) Basidiomycetes
b. Penicillium (ii) Deuteromycetes
c. Ustilago (iii) Ascomycetes
d. Trichoderma (iv) Oomycetes
1. a(iii), b(ii), c(iv), d(i)
2. a(iv), b(iii), c(ii), d(i)
3. a(iii), b(i), c(iv), d(ii)
4. a(iv), b(iii), c(i), d(ii)
Select odd one with respect to Basidiomycetes
1. Mushroom
2. Smut
3. Rust
4. Morels
Mark the incorrect match
1. Saddle joint - Between carpal and metacarpal of the thumb
2. Ball and socket - Between humerus and pectoral girdle
3. Gliding joint - Between carpals
4. Cartilaginous joint - Between prezygapophyses and postzygapophyses of two adjacent vertebrae
UTRs are required for efficient translation, which are present on
1. 3' mRNA
2. 5' mRNA
3. TC region of tRNA
4. Both (1) & (2)
Mark the feature which is not associated with white muscle fibers
1. Lighter in color
2. These muscles have a fast rate of contraction but for a short period
3. They depend mainly on glycolysis for energy production and soon get fatigued
4. Less sarcoplasmic reticulum compared to red muscle fibres
Which of the following plant variety is resistant to yellow mosaic virus?
1. Himgiri
2. Pusa Komal
3. Prabhani kranti
4. Pusa swamim
Constipation is a condition in which defecation occurs only at prolonged intervals. It is irregular or inadequate defecation. Which of the following condition is not responsible for constipation?
1. Decreased colon motility
2. A diet containing too little fluid and fiber
3. Hemorrhoids
4. Too much absorption of water by coion
Taste buds for tasting chilies, Black pepper, and hot sauces are present in
1. Vallarta papillae
2. Fungiform on the upper side
3. Fillon
4. No taste buds for chilies, black pepper, and hot sauces
Mark the incorrect statement with respect to reflex action
1. Response in the form of reflex action immediately protects the animal from harmful situations
2. It relieves the brain from too much strain
3. Opening of mouth on hearing a loud sound is an example of conditioned reflex
4. Interneurons are absent from the monosynaptic reflexes
Which of the following plant must be exposed to light for a period less than a well defined critical photoperiod for flowering in them ?
1. Rice, coffee
2. Oat, radish
3. Cucumber, tomato
4. Cotton, maize
The limbic system controls all the functions except
1. Emotional behaviours expressed as joy, sorrow, liking, disliking
2. Control of food habits necessary for the survival of the individual
3. Control of sexual behaviour necessary for survival of species
4. Regulation of reflexes like coughing, sneezing, swallowing
Which of the following statements about enzymes is false?
1. Enzymes are catalyst within the cells
2. All the cells of an organism contain same enzymes
3. Enzymes bring the substances together so that they undergo a reaction
4. Enzymes lower the activation energy of spontaneous reactions
Which of the following structures of ear provides sensory information on the position of the head in space, is essential for maintaining posture and balances, and also detects linear acceleration and deceleration?
1. Maculae
2. Cristae
3. Semicircular canals
4. Organ of Corti
Amphibians never succeeded in becoming fully terrestrial because amphibian eggs must be laid in water to avoid drying out. Why?
(1) This is because amphibians are anamniotes
(2) Amphibians are amniotes
(3) Amphibians lack allantois
(4) Amphibians lack yolk sac membrane
Just as Aedes is to Dengue
1. Culex is to Filaria
2. Phlebotomus is to Leishmania
3. Trypanosoma is to sleeping sickness
4. Leishmania denovani is to Kala azar
Mycobiont component of lichen provides
1. Water and food materials to phycobiont
2. Organic food to its partner
3. Shelter to algal partner
4. More than one option is correct
The calyptra of capsule has 10 chromosomes in Marchantia. What would be the number of chromosome in its elater?
1. 10
2. 20
3. 30
4. 5
Which of the following is not the feature of facilitated transport?
1. Requires special membrane proteins
2. Highly selective
3. Transport saturates
4. Uphill movement
All given structures of Pinus life cycle are haploid, except
1. Endosperm
2. Pollen
3. Egg
4. Nucellus
Adrenaline or epinephrine hormone is formed from which amino acid?
1. Tyrosine
2. Glycine
3. Tryptophan
4. Alanine
Which of the following is incorrect regarding the hormones, its sources, and its functions?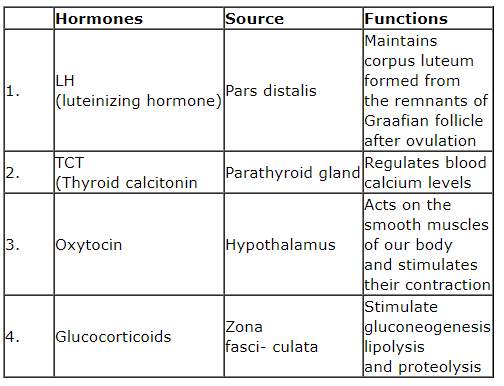
Which receptors are located deep in the dermis and respond to strong pressure and are also sensitive to vibrations?
1. Merkel's discs
2. Meissner's corpuscles
3. Pacinian corpuscles
4. Ruffini's corpuscles
Eleven membered cyclic oligopeptides obtained from fungus Trichoderma polysporum and used as immunosuppressive agent is
1. statin
2. streptokinase
3. cyclosporin A
4. proteases
The effective biocontrol agent for soil-borne diseases in plants is
1 Bacillus thuringiensis
2. Ladybird beetle
3. Trichoderma
4. Baculovirus
Which of the following is a common feature between human sperm and ovum?
1. The same number of mitochondria
2. Presence of zona pellucida outer to the plasma membrane
3. 23 chromosomes
4. Self mostly
The first sign of growing fetus is
1. Movement of foetus
2. Listening to the heart sound of the foetus
3. Morning sickness of the female
4. Detection of HCG in urine
Mark out the correct statement regarding the characteristics of variants in a mixed population
1. Industrialization results in the increase in the types of variants
2. In a mixed population, those that can better adapt, survive and increase in population size
3. Less adapted variant will be completely wiped out
4. All of these
Pouched mammals are present in Australia only due to
1. Lack of placental mammals
2. Due to continental drift pouched mammals in Australia survived
3. Lack of competition from any other mammals
4. Both (2) & (3)
Which one of the following was not advocated by the Darwinian Theory of Evolution?
1. Branching descent
2. Natural selection
3. Mutation caused speciation
4. Fitness is the end result of the adaptive adaptability
in which one of the following options the two examples are incorrectly matched with their particular type of immunity?
Examples Type of immunity
1. Saliva in the mouth - Physiological
and tears in eyes
2. Skin, HCl in the stomach - Physical barriers
and mucus coating
3. Polymorphonuclear - Cellular barriers
leukocytes and
monocytes
4. anti-tetanus and - passive immunity
antisnake bite
injection
Foetus receive some antibodies from the mother through the placenta during pregnancy. This is an example of
1. Naturally acquired active immunity
2. Artificially acquired passive immunity
3. Naturally acquired passive immunity
4. Artificially acquired active immunity
Which of the following statements is correct regarding and transport in blood?
1. 100ml of oxygenated blood carries 53ml of
2. 100ml of blood always transports equal amounts of and
3. 100ml of oxygenated blood delivers morethan to the tissues
4 and are both maximally transported through the blood plasma
Which of the following is incorrect statement about Erythrocyte sedimentation rate(ESR)?
1. It is abnormally enhanced in diseases with abnormal globulin ratio in the plasma
2. ESR is a diagnostic value for many diseases including tuberculosis
3. It gives the relative volume of erythrocytes as the percentage of the blood volume
4. It can be measured by wintrobe and Westergren method
Which of the following is true for QRS complex in an ECG?
1. One can determine the heart beat by counting the number of QRS
2. Represents end of ventricular depolarisation
3. Lubb sound is detected by ECG after QRS complex
4. All of these
Which of the following is incorrect for a one-word scientific term and its description?
1. Aquaculture - Involves the production of
useful aquatic plants and
animals by proper utilization
of small and large water bodies
2. Pisciculture - Production of fishes
3. Blue revolution - Increase in fish production
4. Fishery - Is an industry devoted to catching
processing or selling of fish, shellfish
or other aquatic plants and animals
Mark the wrong statement with respect to medical termination of pregnancy (MTP)
1. 45 to 50 million MTPs are performed each year all over the world
2. It accounts for termination of th of the total number of conceived pregnancies per year
3. It is mainly performed for the purpose of decreasing the population size
4. MTPs are relatively safe during the first trimester of pregnancy
The purification of the biotechnological product is done
1. Upstream processing
2. Downstream processing
3. Thawing processing
4. Enzyme-linked sorbent assay
Ti plasmid of Agrobacterium tumefaciens when disarmed can be used to
1. Induce pest resistance in plants, by being used as a vector
2. Reduce tolerance to abiotic stresses in plants
3. Increase demand of mineral in plants
4. Increase opine production in host plants
In which of the following the larvae are bilaterally symmetrical and the adult are radially symmetrical?
(1) Cnidaria
(2) Echinodermata
(3) Pleurobrachia
(4) Mollusca
Which of the following is not an advantage of transgenic plants?
1. Pest resistant
2. Production of superweeds tolerant to herbicides
3. More tolerant to abiotic stress
4. Prevent early exhaustion of soil fertility
Which of the following is an incorrect statement?
1. 'Bt' in 'Bt-cotton indicates that it is a biotechnology
2. Bt - proteins kill certain insects such as lepidopterans, coleopterans, and dipterans
3. Inactive Bt-prototoxins are converted into active toxins in the alkaline medium in the gut of the insect
4. Bt-toxin is coded by a gene named cry
In artificial insemination technique, the semen is collected
1. Only from the husband and is artificially introduced into the vagina
2. Only from a healthy donor and is artificially introduced inti vagina
3. Either from the husband or from a healthy donor and is artificially introduced only into the vagina
4. Either from the husband or a healthy donor is artificially introduced into the vagina or into the uterus
Which one of the following statement is incorrect ?
1. The generative cell divides during the growth of pollen tube in th stigma
2. In some species floral rewards are in providing safe places to lay eggs
3. The embryo development precedes endosperm development
4. Scutellum is the thin large cotyledon in monocots.
Which one of the following flowers produce assured seed-set even in the absence of pollinators ?
1. Autogamous
2. Homogamous
3. Cleistogamous
4. Herkogamous
Cross pollination is encouraged if
1. Flower is self fertile
2. Homogamy is present
3. Flower is diocious
4. Flowers are hermaphrodite
Aleurone layer has same ploidy as that of
1. PEN
2. Zygot
3. Pericarp
4. Scutellum
in a particular climatic condition, decomposition rate is slower if detritus is rich in
1 nitrogen and water-soluble substances
2 chitin and nitrogen
3 lignin and chitin
4 water-soluble substances and lignin
Mark correct match
Column I Column II
a. Thick and swollen cotyledons (i) Castor
b. Unused endosperm in mature seed (ii) Legumes
c. Unused nucellus in seed (iii) Cashewnut
d. Thalamus contributes to fruit formation (iv) Beet
1. a(iii), b(iv), c(ii), d(i)
2. a(i), b(ii), c(iv), d(iiii)
3. a(ii), b(i), c(iii), d(iv)
4. a(ii), b(i), c(iv), d(iii)
MacArthur found that closely related species of warblers living on the tree shows
1. competitive exclusion
2. coexistence
3. interference competition
4. competition release
A perennial stem, which generally lies horizontally below the surface, is an organ for propagation in
1. Ginger
2. Potato
3. Colchicum
4. Allium
Under which of the following conditions, appreciable amount of glucose may appear in the urine?
A. Diabetes mellitus not controlled.
B. Glucose concentration in the blood plasma is above 180 mg/di
C. Glucose concentration of the blood exceeds the renal threshold value of glucose
D. Diabetes inspidus
1. A and D
2. Only A and C
3. A, B, and C
4. Only B and C
Match column l with column ll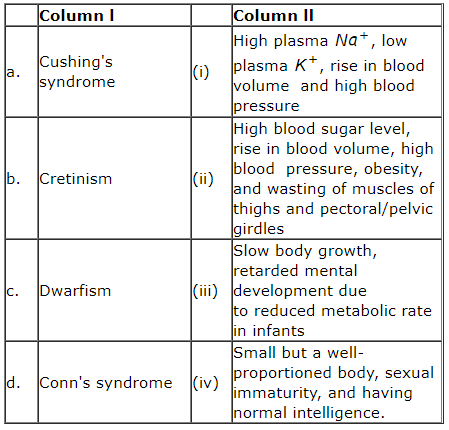
1. a(ii), b(iii), c(iv), d(i)
2. a(ii), b(iv), c(iii), d(i)
3. a(i), b(iii), c(iv), d(ii)
4. a(iv), b(iii), c(iii), d(i)
Which of the following substances is normally not excreted during urine formation?
1.
2.
3. Ammonia
4. Glucose
Which of the following is always sterile?
a. Triploid
b. Amphidiploid
c. Autotetrapolyploid
d. Allotetrapolyploid
1. A and D only
2. B and C only
3. A only
4. D only
Following figure represents which type of chromosomal aberration?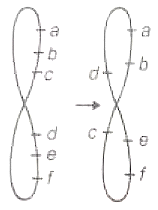
1. Deletion
2. Duplication
3. Paracentric inversion
4. Pericentric inversion
There are certain limitations of ecological pyramids, like
1. It does not take into account the same species belonging to two or more trophic levels
2. It assumes a long food chain of many trophic levels
3. saprobic organisms are not given any place
4. More than one option is correct
Which one of the following is matched correctly?
1. low productive ecosystem - Flood plains
2. Average productive ecosystem - Grassland
3. least productive ecosystem - Temperate forest
4. High productive ecosystem - Area of upwelling
What will be the genotype of proposita in the family if following pedigree chart is given?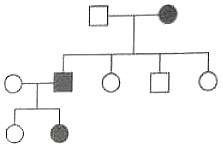
1. AA
2. Aa
3.
4.
Select a incorrect statements regarding pyramids
1. These are proposed by Charles Elton
2. It does not explain the role of organisms present at more than two trophic levels
3. Decomposers are not given importance while preparing a pyramid
4. It is sometimes upright, if prepared for energy
Interaction of two genes in which one gene has no effect when alone but it modifies the effect of another gene when present in dominant form, is called ______ interaction
1. Inhibitory gene
2. Dominant epistatic
3. Duplicate
4. Supplementary
Which of the following statements is depicted by the floral formula - 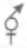 ?
?
1. Number of calyx is four arranged in single whori
2. It represents family Brassicaceae
3. Adhesion of carpels
4. Clawless crucifer corolla
Both the alleles produce their effect in
1. Heterozygous B blood group
2. Blood group AB
3. Snapdragon flower colour
4. Cucurbits fruit colour
If a hybrid DNA molecule labelled with is allowed to replicate four times in culture medium having , the hybrid DNA after third generation would be
1. 50%
2. 25%
3. 12.5%
4. 6.5%
What is incorrect for DNA?
1. Radius for DNA is 1 nm
2. Phosphate moiety is at 5'-end of deoxyribose sugar
3. Nitrogen bases linked to sugar moiety project inward
4. Distance between two successive base pair strands is 3.4 nm
The most common type and advanced type of placentation is found respectively in
1. Solanaceae and Poaceae
2. Leguminosae and Asteraceae
3. Brassicaceae and Liliaceae
4. Asteraceae and Liliaceae
Nitrogen bases present in the introns at 5' in the beginning and at 3' in the last are
1. AG and GU
2. AU and GU
3. GU and AG
4. GU and AU
Who proposed semiconservative mode of replication in DNA ?
1. Meselson and Stahl
2. Watson and Crick
3. Cairn
4. Taylor
Collenchyma is characterised by
1. Absence of protoplast at maturity
2. Supporting young stem and leaf petiole
3. Lignocellulosic thickening in secondary walls
4. Common presence of intercellular spaces
Phellogen is a kind of secondary meristem
1. Cutting phelloderm outside and phellum inside
2. Usually developing in the cortex region of dicot stem
3. Forming non-suberised cells outside
4. Made of narrow, thick walled, suberised cells
Which of the following statement is not related to centriole?
1. Made up of nine evenly spaced peripheral fibrils of tubulin protein
2. Spindle fibres that give rise to spindle apparatus during cell division in plant cells
3. Central part of the proximal region is called the hub
4. It form the basal body of eukaryotic flagella
A number of organised flattened membranous sacs are present in the
1. Matrix of mitochondria
2. Nucleoplasm
3. Chloroplasts
4. Matrix of centriole
Membrane bound vesicular structure formed by the process of packaging in the Golgi apparatus
1. Contains materials not useful for cell and is bounded by half unit membrane
2. Contains all types of hydrolytic enzymes optimally active at the acidic pH
3. Are concerned with photorespiration in plants and animals
4. Possess enzymes for Krebs cycle
Pseudostratified nonciliated columnar epithelium tissue is found in
(1) Urethra of male
(2) Linning of trachea
(3) Epididymis
(4)Proximal tubules of kidney
Which of the following statement is incorrect?
(1) The axon terminals may form synapses with dendrite terminals, cell bodies or even axons of other neurons
(2) Nissi bodies are absent in axon and axonal hillock
(3) Nervous tissue is made of neurons and neuroglia cells
(4) The neurotransmitter is always released from dendrite part of a neuron
Nitrogen fixing ability is found in
(1) Some anaerobic microbes only
(2) Some aerobic microbes only
(3) Some eubacteria and archaebacteria also
(4) Some photosynthetic and chemosynthethic microbes only
In hydroponics
(1) Cost of infrastructure is low
(2) Soil borne pathogens are absent
(3) Problem of weeding is present
(4) pH is maintained at 7-8
First stable product of PCR cycle is
(1) Oxaloacetic acid
(2) Sedoheptulose-1, 7-diphosphate
(3) 3-phosphoglyceraldhyde
(4) 3-phosphoglyceric acid
Some self feeder bacteria show anoxygenic photosynthesis and lack pigment system II. This statement proves that
(1) Splitting of is coupled to PS-I
(2) PS-1 developed earlier in evolution
(3) PS-II developed earlier in evolution
(4) Splitting of occurs only in eukaryotes
During glycolysis how many redox equivalents are removed from PGAL to form a reduced molecule of co-enzyme?
1. One
2. Two
3. Three
4. Four
How many NADH molecules are oxidised to with the help of ETC, if one molecule of glucose is oxidised completely inside the nerve cell?
(1) 10
(2) 8
(3) 12
(4) 2
Alkyl cyanide \(R-C \equiv N\) and alkyl isocyanides \(R- \overset{+}{N} \equiv C^-\) are:
| 1. | Tautomers | 2. | Metamers |
| 3. | Functional isomers | 4. | Geometrical isomers |
The optically active compound among the following is-
1. Isobutyric acid
2. beta-Chloropropionic acid
3. Propionic acid
4. alpha-Chloropropionic acid
Which among the given molecules can exhibit tautomerisrn?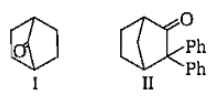
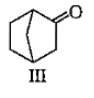
1. III Only
2. Both I and III
3. Both Iand II
4. Both II and III
In the Kjeldahl's method for estimation of nitrogen present in a soil sample, ammonia evolved from 0.75 g of sample neutralised 10 mL of 1 M H2SO4. The percentage of nitrogen in the soil is
1. 37.33 2. 45.33
3. 35.33 4. 43.33
The ionisation constant of ammonium hydroxide is 1.77 x 10-5 at 298 K. Hydrolysis constant of ammonium chloride is
1. 5.65 x 10-10
2. 6.50 x 10-12
3. 5.65 x 10-13
4. 5.65 x 10-12
The pOH of a solution at 25C that contains 1 x 10-10 M of hydronium ions is:
1. 7.00
2. 4.00
3. 9.00
4. 1.00
The osmotic pressure of 5 % (mass-volume) solution of cane sugar at 150 °C (mol. mass of sugar = 342 g/mole) is:
| 1. | 4 atm | 2. | 5.07 atm |
| 3. | 3.55 atm | 4. | 2.45 atm |
A solution containing 6.8 g of a non-ionic solute in 100 g of water was found to freeze at −0.93 oC. The freezing point depression constant of water is 1.86. The molecular weight of the solute is-
| 1. | 13.6 m | 2. | 34 m |
| 3. | 68 m | 4. | 136 m |
The rate constant for a reaction of zero-order in A is 0.0030 mol L-1 s-1. How long will it take for the initial concentration of A to fall from 0.10 M to 0.075 M?
1. 8.3 sec
2. 0.83 sec
3. 83 sec
4. 10.3 sec
The process requiring the absorption of energy is :
1.
2.
3.
4.
Which among the following is incorrect?
| Complex compounds | Type of hybridization |
| 1. [V(NH3)6]3+ | d2sp3 |
| 2. [CrCl3(NMe3)3] | d2sp3 |
| 3. [Cu(CN)(NO2)(NH3)(py)] | dsp2 |
| 4. K3[Co(ox)3] | sp3d2 |
The incorrect option among the following is:
| 1. | [Co(NO2)(H2O)(en)2]Cl2, [CoCl(NO2)(en)2]Cl·H2O | Hydrate isomerism |
| 2. | [Cu(NH3)4[PtCl4], [CuCl(NH3)3][PtCl3(NH3)] | Co-ordination isomerism |
| 3. | [Ni(CN)(H2O)(NH3)4]Cl, [NiCl(H2O)(NH3)4]CN | Ionization isomerism |
| 4. | [Cr(NO2)(NH3)5][ZnCl4], [Cr(NO3)(NH3)5][ZnCl4] | Linkage isomerism |
In a reaction, the rate = k[A]1[B]-2/3 the order of the reaction is-
1. 1/3
2. 2
3. -1/3
4. Zero
The rate of reaction triples when the temperature changes from \(20{ }^{\circ} \mathrm{C} \text { to } 50^{\circ} \mathrm{C}\). The energy of activation for the reaction will be:
| 1. | \(28.81 \mathrm{~kJ} \mathrm{~mol}^{-1} \) | 2. | \(38.51 \mathrm{~kJ} \mathrm{~mol}^{-1} \) |
| 3. | \(18.81 \mathrm{~kJ} \mathrm{~mol}^{-1} \) | 4. | \(8.31 \mathrm{~kJ} \mathrm{~mol}^{-1}\) |
The correct order of covalent bond character among
LiCl, BeCl2, BCl3 and CCl4, is:
1. LiCl < BeCl2 > BCl3 > CCl4
2. LiCl > BeCl2 < BCl3 < CCl4
3. LiCl < BeCl2 < BCl3 < CCl4
4. LiCl > BeCl2 > BCl3 > CCl4
The density of a gaseous substance at 1 atm pressure and 773 K is 0.4 g/L. If the molecular weight of the substance is 30, the dominant force existing among gas molecules is/are-
| 1. | Repulsive | 2. | Attractive |
| 3. | Both (1) & (2) |
4. | None of these |
Van der Waals equation at low pressure is -
1. PV = RT - Pb
2. PV = RT -
3. PV = RT +
4. PV = RT + Pb
A 0.1 molar solution of weak base BOH is 1% dissociated. If 0.2 moles of BCl is added in 1 litre solution of BOH, the degree of dissociation of BOH will become
1.
2.
3. 0.05
4. 0.02
The product of acetone with is
1.
2.
3.
4.
The final product Y in the below mentioned sequence is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Consider the following reaction
The structure of B is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The products A and B in the below mentioned reaction are, respectively:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
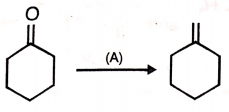
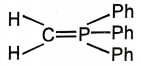
This conversion may be carried by using reagent A.
A maybe
1. 
2.
3.
4.
The numbers of lone pairs and bond pairs in hydrazine are, respectively
1. 2 and 4
2. 2 and 6
3. 2 and 5
4. 1 and 5
Oxidation states of C in Toluene is/are
1. -3
2. -1
3. 0
4. All of these
As an isolated box, equally partitioned, contains two ideal gasses A and B as shown:
When the partition is removed, the gases mix. The changes in enthalpy and entropy in the process, respectively, are
1. Zero, positive
2. Zero, negative
3. Positive, zero
4. Negative, zero
The for vaporization of a liquid is \(20 \mathrm{~kJ} / \mathrm{mol}.\) Assuming ideal behaviour, the change in internal energy for the vaporization of \(1 \mathrm{~mol}\) of the liquid at \(60^{\circ} \mathrm{C}\) and 1 bar is close to:
| 1. | \(13.2 \mathrm{~kJ} / \mathrm{mol} \) | 2. | \(17.2 \mathrm{~kJ} / \mathrm{mol} \) |
| 3. | \(19.5 \mathrm{~kJ} / \mathrm{mol} \) | 4. | \(20.0 \mathrm{~kJ} / \mathrm{mol}\) |
The amount of required to completely reduce 100 mL of 0.25 N iodine solution, is
1. 6.20 g
2. 9.30 g
3. 3.10 g
4. 7.75 g
A metal Oxide can be reduced by hydrogen to give free metal and water. O. 1596 gm of metal Oxide requires 6 mg Of hydrogen for complete reduction. The atomic weight of the metal is:
1. 27.9
2. 159.6
3. 79.8
4. 55.8
| Molar mass | |
| 461 |
The compound that does not reduce Benedict's solution is:
| 1. | Glucose | 2. | Fructose |
| 3. | Sucrose | 4. | Aldehyde |
A photon with an initial frequency of \(10^{11}~\mathrm{Hz}\) scatters off an electron at rest. Its final frequency is \(0.9 \times10^{11}~\mathrm{Hz}.\) The speed of scattered electron is close to:
| 1. | \(3 \times10^{2}~\mathrm{ms}^{-1}\) | 2. | \(3.8 \times10^{3}~\mathrm{ms}^{-1}\) |
| 3. | \(2 \times10^{6}~\mathrm{ms}^{-1}\) | 4. | \(30~\mathrm{ms}^{-1}\) |
The wavelength of a certain line in the Balmer series is observed to be 4341 Ao for hydrogen atoms.
The electronic transition among the following may be:
1. 3 → 2
2. 4 → 1
3. 5 → 2
4. 5 → 3
Zieglar-Natta catalyst is
1.
2.
3.
4.
A metal ion has configuration. In octahedral complex entity of low spin, the number of electrons present in set of orbitals will be
| 1. | 6 | 2. | 4 |
| 3. | 0 | 4. | 2 |
A compound is made by mixing cobalt (III) nitrite and potassium nitrite solution in a ratio of 1 : 3. The aqueous solution of the compound showed 4 particles per molecule whereas molar conductivity reveals the presence of six electrical charges. The formula of the compound is:
1. Co(NO2)3 · 2KNO2
2. Co(NO2)3 · 3 KNO2
3. K3[Co(NO2)6]
4. K[Co(NO2)4]
Gypsum on heating can change to :
1. Orthorhombic form.
2. Plaster of paris.
3. Dead plaster.
4. All of the above.
Out of the following which is a hydride?
1. Hydrolith
2. Oxidane
3. Azane
4. All of these
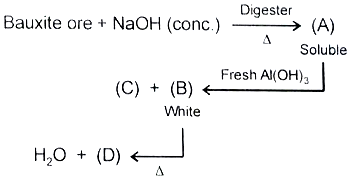
Compound (D) is
1.
2.
3.
4.
NaCN is a depressant that is used:
| 1. | To Separate PbS from ZnS by forming a complex with PbS. |
| 2. | To Separate PbS from ZnS by forming a complex with ZnS. |
| 3. | To Form froth. |
| 4. | As a collector. |
While Fe3+ is stable, Mn3+ is not stable in acidic solution because
1. O2 oxidises Mn2+ to Mn3+
2. O2 oxidises both Mn+2 to Mn+3 and Fe2+ to Fe3+
3. Fe3+ oxidises H2O to O2
4. Mn3+ oxidised H2O to O2
The lanthanide compound which is used as a most powerful liquid laser after dissolving in selenium oxychloride is
1. Cerium oxide
2. Neodymium oxide
3. Promethium sulfate
4. Cerium sulfate
Equivalent conductance of saturated \(\mathrm {BaSO}_4\) solution is \(400 \mathrm { ~ohm}^{-1}\) \(\mathrm {cm}^2\) \(\mathrm { ~equivalent}^{-1}\) and it's specific conductance is \(8 \times 10^{-5} \text { ohm}^{-1} \text {cm}^{-1}\) ; hence solubility product \(K_{sp}\) of \(\mathrm {BaSO}_4\) is :
1. \(4 \times 10^{-8} \text {M}^2\)
2. \(1 \times 10^{-8} \text {M}^2\)
3. \(2 \times 10^{-4} \text {M}^2\)
4. \(1 \times 10^{-4} \text {M}^2\)
A diatomic molecule has a dipole moment of 1.2 D. If its bond length is equal to 10 -10 m then the fraction of an electronic charge on each atom will be:
1. 42%
2. 52%
3. 37%
4. 25%
The reddish-brown precipitate formed in the Fehling's test for aldehydes (RCHO) is due to the formation of
1. Cu
2.
3. CuO
4.
Some equipotential surfaces are shown in the figure. The electric field at points \(A\), \(B\) and \(C\) are respectively:

| 1. | \(1~\text{V/cm}, \frac{1}{2} ~\text{V/cm}, 2~\text{V/cm} \text { (all along +ve X-axis) }\) |
| 2. | \(1~\text{V/cm}, \frac{1}{2} ~\text{V/cm}, 2 ~\text{V/cm} \text { (all along -ve X-axis) }\) |
| 3. | \(\frac{1}{2} ~\text{V/cm}, 1~\text{V/cm}, 2 ~\text{V/cm} \text { (all along +ve X-axis) }\) |
| 4. | \(\frac{1}{2}~\text{V/cm}, 1~\text{V/cm}, 2 ~\text{V/cm} \text { (all along -ve X-axis) }\) |
If \(\overrightarrow{A} \times \overrightarrow{B} = \overrightarrow{C} + \overrightarrow{D}\), then which of the following statement is correct?
| 1. | \(\overrightarrow B\) must be perpendicular to \(\overrightarrow C\) |
| 2. | \(\overrightarrow A\) must be perpendicular to \(\overrightarrow C\) |
| 3. | Component of \(\overrightarrow C\) along \(\overrightarrow A\) = Component of \(\overrightarrow D\) along \(\overrightarrow A\) |
| 4. | Component of \(\overrightarrow C\) along \(\overrightarrow A\) = - (Component of \(\overrightarrow D\) along \(\overrightarrow A\)) |
For a dipole, q = 2 x 10–6 C and d = 0.01 m, if E = 5 x 105 N/C, then the maximum torque for this dipole is:-
1.
2.
3.
4.
The position of a particle is given by , momentum . The angular momentum is perpendicular to :-
1. X-axis
2. Y-axis
3. Z-axis
4. Along the line at equal angles to all the three axes
A positive charge q is placed in front of a conducting solid cube at a distance d from its centre. Find the electric field at the centre of the cube due to the charges appearing on its surface:-
(1) , towards the charge q
(2) , ,away from charge q
(3) zero
(4) Can’t be found
| 1. | plane parallel to \(yz\) plane |
| 2. | plane parallel to the \(x\text{-}\)axis |
| 3. | concentric circle centered at the origin |
| 4. | coaxial cylinder with axis parallel to the \(y\text-\)axis |
A resistor R1 dissipates the power P when connected to a certain generator. If the resistor R2 is put in series with , the power dissipated by R1:-
(1) Decreases
(2) Increases
(C) Remains the same
(D) Any of the above depending upon the relative values of R1 and R2
The graph below shows position as a function of time for two trains running on parallel tracks.
Which of the following statements is true?
| 1. | At time \(t_B \) both the trains have the same velocity |
| 2. | Both the trains have the same velocity at some time after \(t_B \) |
| 3. | Both the trains have the same velocity at some time before \(t_B \) |
| 4. | Both the trains have the same acceleration |
Find the equivalent resistance between A and E (the value of each resistor is R).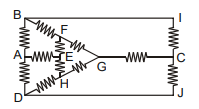
(1)
(2)
(3)
(4)
To the captain of a ship \(A\) travelling with velocity \(\overrightarrow{v_{A}} = \left( 3 \hat{i} - 4 \hat{j} \right)\) km/h, a second ship \(B\) appears to have a velocity \(\overrightarrow{v_{B}} = \left(5 \hat{i} +12 \hat{j} \right)\) km/h. What is the true velocity of the ship \(B\)?
1. \(2 \hat{i} + 16 \hat{j}\) km/h
2. \(13 \hat{i} + 8 \hat{j}\) km/h
3. \(- 2 \hat{i} - 16 \hat{j}\) km/h
4. none of these
A varying horizontal force F = at acts on a block of mass m kept on a smooth horizontal surface. An identical block is kept on the first block. The coefficient of friction between the blocks is µ. The time after which the relative sliding between the blocks prevails is:-
1.
2.
3.
4.
A circuit has section \(AB\) as shown in figure. The emf of the cell is \(10\) V. The potential difference \(V_{AB}= 5~\text{V}\). The charge on the capacitor \(C_1\) is:

| 1. | \(10~\mu\text{C}\) | 2. | \(5~\mu\text{C}\) |
| 3. | \(15~\mu\text{C}\) | 4. | Can't be determine |
A body has an initial velocity of 3 m/s and has an acceleration of 1 m/s2 normal to the direction of the initial velocity. Then its velocity 4 seconds after the start is:
(1) 7 m/s along the direction of initial velocity
(2) 7 m/s along the normal to the direction of initial velocity
(3) 7 m/s mid-way between the two directions
(4) 5 m/s at an angle of tan–1 (4/3) with the direction of initial velocity
A chain of length L is placed on a horizontal surface as shown in figure. At any instant x is the length of chain on rough surface and the remaining portion lies on smooth surface. Initially x = 0. A horizontal force P is applied to the chain (as shown in figure). In the duration x changes from x = 0 to x = L, for chain to move with constant speed-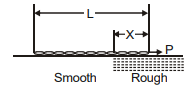
1. the magnitude of P should increase with time
2. the magnitude of P should decrease with time
3. the magnitude of P should increase first and then decrease with time
4. the magnitude of P should decrease first and then increase with time
In a square loop made with a wire of uniform cross-section current I enters from point A and leaves from point B. The magnetic field strength at the centre of the square is :-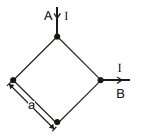
1. Zero
2.
3.
4.
A ferromagnetic substance is placed in the varying magnetising field \(H.\) The magnetic induction \(B\) is measured for various values of direct and reverse magnetising fields. The following graph has been plotted for \(B\) versus \(H.\) Choose the wrong statement:
| 1. | there is a limit of direct and reverse external magnetising field at which the magnetisation and hence the magnetic induction saturates. |
| 2. | even after removing the external magnetising field some residual magnetisation called 'retentivity' is left over in the substance. |
| 3. | on increasing the reverse magnetising field, the magnetic field decreases to zero for a value of magnetising field which is known as 'susceptibility'. |
| 4. | on increasing the reverse magnetising field, the magnetic field decreases to zero for a value of magnetising field known as 'coercivity'. |
1. \(u^{2} \sin^{2}\alpha\)
2. \(\dfrac{m u^{2} \cos^{2} \alpha}{2}\)
3. \(\dfrac{m u^{2}\sin^{2} \alpha}{2}\)
4. \(- \dfrac{m u^{2}\sin^{2} \alpha}{2}\)
The system shown in figure is released from rest with mass 2 kg in contact with the ground. Pulley, spring and strings are massless and friction is absent everywhere. The speed of 5 kg block when 2 kg block leaves the contact with the ground is (force constant of the spring, k = 40 Nm–1 and g = 10 ms–2):-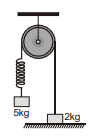
(1)
(2)
(3)
(4)
A square loop of side 'a' hangs from an insulating hanger of spring balance. The magnetic field of strength B occurs only at the lower edge of loop which carries a current I. Find the change in the reading of the spring balance if the direction of current is reversed :-
1. IaB
2. 2IaB
3.
4.
A 1.0 m long metallic rod is rotated with an angular frequency of 400 rad s–1 about an axis normal to the rod passing through its one end. The other end of the rod is in contact with a circular metallic ring. A constant and uniform magnetic field of 0.5 T parallel to the axis exists everywhere. The emf developed between the centre and the ring is:-
(1) 10 V
(2) 1 V
(3) 100 V
(4) 200 V
A straight solenoid has \(50\) turns per cm in primary coil and \(200\) turns in the secondary coil. The area of cross-section of the solenoid is \(4\) cm2. Calculate the mutual inductance.
1. \(5.0~\text{H}\)
2. \(5.0\times 10^{-4}~\text{H}\)
3. \(2.5~\text{H}\)
4. \(2.5\times 10^{-4}~\text{H}\)
A man hangs from a rope attached to a hot-air balloon. The man's mass is greater than the mass of the balloon and its contents. The system is stationary in the air. If the man now climbs up to the balloon using the rope, the centre of mass of the "man plus balloon" system will:
| 1. | remain stationary |
| 2. | move up |
| 3. | move down |
| 4. | first moves up and then return to its initial position |
A loss-free transformer having 100 turns in primary is used to transmit 10 kW of power. The input voltage is 200 V and power is transmitted at 5 kV. The currents in the primary and secondary coils of the transformer are-
(1) 2 A and 50 A
(2) 50 A and 2 A
(3) 25 A and 4 A
(4) 12.5 A and 8 A
A ring of mass m and radius r is released from rest along a smooth inclined plane of angle of inclination . The angular momentum of the body about the instantaneous point of contact after a time t from the instant of release is equal to
(1) mgrt cos
(2) mgrt sin
(3) (3/2) mgrt sin
(4)
A mass m such that m=M is supported by a massless string wound round a uniform cylinder of mass M and radius R. On releasing the mass from rest, it will fall with acceleration:-
1. g
2.
3.
4.
Three rods made of the same material, having the same cross-sectional area but different lengths 10 cm, 20 cm and 30 cm are joined as shown. The temperature of the junction will be:-
1. \(10.8^{\circ}\mathrm{C}\)
2. \(14.6^{\circ}\mathrm{C}\)
3. \(16.4^{\circ}\mathrm{C}\)
4. \(18.2^{\circ}\mathrm{C}\)
If at a pressure of \(10^6\) dyne/cm2, one gram of nitrogen occupies \(2\times10^4\) c.c. volume, then the average energy of a nitrogen molecule in erg is:
| 1. | \(14\times10^{-13}\) | 2. | \(10\times10^{-12}\) |
| 3. | \(10^{6}\) | 4. | \(2\times10^{6}\) |
If the temperature of the source is increased, then the efficiency of a Carnot engine:-
1. Increases
2. decreases
3. Remains constant
4. First increases and then remains constant
A geyser heats water flowing at the rate of 3.0 litre per minute from 27°C to 77°C. If the geyser operates on a gas burner and its heat of combustion is 4.0 × 104 J/g, then what is the rate of combustion of fuel (approx) ?
(1) 24 gm/min
(2) 12 gm/min
(3) 32 gm/min
(4) 16 gm/min
The emission spectrum of a black body at two different temperatures are shown by curves P and Q in figure. The ratio of the areas under the two curves P and Q will be :-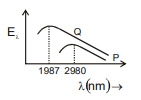
(1) 1 : 16
(2) 4 : 19
(3) 81 : 256
(4) 16 : 81
A mark on the surface of the sphere \(\left(\mu= \frac{3}{2}\right)\) is viewed from a diametrically opposite position. It appears to be at a distance \(15~\text{cm}\) from its actual position. The radius of the sphere is:
1. \(15~\text{cm}\)
2. \(5~\text{cm}\)
3. \(7.5~\text{cm}\)
4. \(2.5~\text{cm}\)
In an astronomical telescope, the focal length of the objective lens is \(100\) cm, and of eyepiece is \(2\) cm. The magnifying power of the telescope for the normal eye is:
1. \(50\)
2. \(10\)
3. \(100\)
4. \(\frac{1}{50}\)
In Young's experiment performed with light of wavelength 6000 Å, when a thin glass plate of refractive index 1.5 is placed in the path of one of the interfering beams, the central bright fringe shifts by 2 mm to a position previously occupied by the seventh bright fringe. Then find thickness of the plate.
1. 10.2
2. 12
3. 6.8
4. 8.4
A container has two immiscible liquids of densites and (> ). A capillary tube of radius r is inserted in the liquid so that its bottom reaches upto the denser liquid. The denser liquid rises in the capillary and attains a height h from the interface of the liquids, which is equal to the column length of the lighter liquid. Assuming angle of contact to be zero, the surface tension of heavier liquid is :-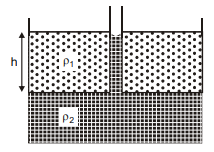
1.
2.
3.
4.
A barometer kept in an elevator reads 76 cm when it is at rest. If the elevator goes up with increasing speed, the reading will be
(1) zero
(2) 76 cm
(3) >76 cm
(4) < 76 cm
Water flows through a horizontal tube as shown in figure. If the difference in heights of water column in vertical tubes is 80 cm and the area of cross-section at A and B are 5 cm2 and 3 cm2 respectively, then the rate of flow of water at A( in cc/sec) is:-
[ ]
1.
2.
3. 150
4. None of these
A radioactive sample decays by 63% of its initial value in 10 s. It would have decayed by 50% of its initial value in :
1. 7 s
2. 14 s
3. 0.7 s
4. 1.4 s
Two satellites S1 and S2 are revolving around a planet in coplanar and concentric circular orbits of radii R1 and R2 in the same direction respectively. Their respective periods of revolution are 1 hr and 8 hr. The radius of the orbit of satellite S1 is equal to 104 km. The relative speed when they are closest will be:
(1)
(2)
(3)
(4)
The kinetic energy of an electron is 5 eV. Calculate the de-Broglie wavelength associated with it (h =6.6 × 10–34 Js, me = 9.1 × 10–31 kg)
1. 5.47
2. 10.9
3. 2.7
4. 12.01
Two pendulums begin to swing simultaneously. The first pendulum makes 9 full oscillations when the other makes 7. The ratio of lengths of the two pendulums is :
(1) 9/7
(2) 7/9
(3) 49/81
(4) 81/49
Suppose an electron is attracted towards the origin by a force where 'k' is a constant and 'r' is the distance of the electron from the origin. By applying Bohr model to this system, the radius of the nth orbital of the electron is found to be 'rn' and the kinetic energy of the electron to be 'Tn'. Then which of the following is true:-
1. independent of n,
2.
3.
4.
From given graph calculate 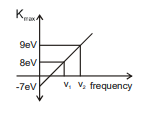
(1)
(2)
(3)
(4)
Logic gates \(X\) and \(Y\) have the truth tables shown below:

| \(X\) | ||
| \(P\) | \(Q\) | \(R\) |
| \(0\) | \(0\) | \(0\) |
| \(1\) | \(0\) | \(0\) |
| \(0\) | \(1\) | \(0\) |
| \(1\) | \(1\) | \(1\) |

| \(Y\) | |
| \(P\) | \(R\) |
| \(0\) | \(1\) |
| \(1\) | \(0\) |
When the output of \(X\) is connected to the input of \(Y\), the resulting combination is equivalent to a single:
1. NOT gate
2. OR gate
3. NAND gate
4. AND gate
In a transistor circuit shown here, the base current is \(35~\mu \)A . The value of the resistor\(R_{b}\) is:
(Given \(V_{BE}=4.5\) V)
1. \(128.5~\mathrm{k\Omega}\)
2. \(257~~\mathrm{k\Omega}\)
3. \(5~~\mathrm{k\Omega}\)
4. \(2.5~~\mathrm{k\Omega}\)
When forward bias is applied to a P–N junction, then what happens to the potential barrier VB and the width of depletion region x ?
1. increases, x decreases
2. decreases, x increases
3. increases, x increases
4. decreases, x decreases