Malleolus is the part of
1. Humerus
2. Femur
3. Ulna
4. Tibia
Which of the following is an incorrect statement with respect to? the axial skeleton of humans?
1. A single U-shaped bone called hyoid bone is present at the base of the buccal cavity and it is included in the skull
2. The skull region articulates with the superior region of the vertebral column with the help of one occipital condyle
3. The sternum is a flat bone present on the ventral midline of thorax
4. Last 2 pairs (11th and 12th) of ribs are not connected ventrally and are therefore called floating ribs
Transduction experiment to prove DNA as a genetic material was conducted by Hershey and Chase with the use of radioactive elements, namely
1.
2.
3.
4.
Choose the odd one out with respect to green manure
1. Reduces soil alkalinity
2. Prevents soil erosion
3. Increases C/N ratio in soil
4. Increases crop yield by 30-50% as compared to farmyard manure
Which of the following hormone inhibits the secretion of gastric juice?
1. Enterogastrone
2. Secretin
3. Cholecystokinin
4. All of these
Brunner's glands are
1. Simple tubular
2. Simple coiled tubular
3. Compound alveolar
4. Compound tubular
Find out the incorrectly matched pair with respect to structure and its function as mentioned below:
| 1. | Eustachian tube | - | Maintains air pressure in the tympanic cavity |
| 2. | Cornea | - | Controls amount of light entering into an eyeball, called the diaphragm of eye |
| 3. | Cristae in ampulla | - | Detect rotational motion |
| 4. | Semicircular canals | - | Help in maintaining dynamic equilibrium, not in hearing |
Stimulation by a parasympathetic nervous system is likely to cause all the following except
1. Increased pancreatic exocrine secretion
2. Increased bile synthesis
3. Decreased insulin secretion
4. Increased gastric secretion
Which of the following is incorrect match about the enzyme and its co-factor?
Enzyme Co-factor
1. Carboxypeptidase : Zinc
2. Tyrosinase : Copper
3. Carbonic anhydrase : Zinc
4. Catalase :
Which layer of the eyeball becomes thick in the anterior part to form a ciliary body?
A. Sclera
B. Choroid
C. Retina
D. Conjunctiva
1. A only
2. B only
3. C only
4. Both A & B
The neurologic disturbances seen in Niemann-pick disease are associated with the accumulation in the central nervous tissue which one of the following?
1. Prostaglandins
2. Ganglioside
3. Cerebroside
4. Sphingomyelin
Which of the following is not a correct statement with respect to the human eyes?
1. When all cones are stimulated equally, a sensation of white light is produced. The sensations of different colors are produced by various combinations of these cones and their photopigments.
2. Fovea of the macula lutea has the greatest visual acuity and resolution
3. Cones produces daylight (Photopic) vision and rods produces twilight (Scotopic) vision
4. Ciliary muscles contract when we focus our eyes on a far-placed object
Which of the following is the most abundant protein in animal tissue?
1. Ribulose diphosphate carboxylase-oxygenase
2. Collagen
3. Trypsin
4. GLUT-4
Which of the following statement is correct about the life cycle of Ascaris lumbricoides?
A. Infective stage - Embryonated egg with II Juvenile
B. Site of location of worm - Small intestine
C. Four moultings of the larva occur two outside in the soil within the egg shell, one in lungs and one in intestine.
D. Pathogenesis - About a hundred to a thousand or even more, upto 5000 worms have been recorded causing blockage of intestine in man
1. A & B
2. A, B, C
3. A, B, C & D
4. A, B & D
Which one of the following is not a matching pair of an animal and a certain phenomenon it exhibits?
1. Obelia : Metagenesis
2. Pila : Torsion
3. Octopus : The animal moves rapidly by jet propulsion mechanism
4. Nereis : Monoecious
Which of the following hormones acts on its target tissues by a steroid hormone-like mechanism of action?
1. Thyroid hormone
2. FSH
3. Glucagon
4. ADH
Hassal's corpuscles are found in
1. Pituitary gland
2. Pineal gland
3. Thymus gland
4. Thyroid gland
Find the incorrect statement is
a flocs are masses of anaerobic bacteria associated with fungal filarnents
b BOD gets reduced significantly when the secondary treatment is done using aerobic microbes
c Activated sludge can be sediment only after anaerobic digestion of sewage
d Mixture of gases is released during aerobic digestion of sewage
1. a, b, c, & d
2. Only (c)
3. All except (b)
4. a, b & d
The target of FSH in males is
1. Granulosa cells
2. Sertoli cells
3. Interstitial cells
4. Both (1) & (2)
Basic principles of embryonic development were described by
1. Von Baer
2. Mayr
3. Darwin
4. Weismann
Term 'Semen' is used for
1. Fluid present in vas deferens
2. Fluid present in seminiferous tubules
3. Fluid present in seminal vesicles
4. Fluid present in membranous urethra
Which of the following extraembryonic membrane forms the human placenta?
a. Allantois
b. Chorion
c. Yolk sac
d. Amnion
e. Vitelline membrane
1. a, b, c, d & e
2. a & b
3. b & d
4. b only
Whales, bats, cheetah, and humans (all mammals) share similarities in the pattern of bones of forelimbs. Though these forelimbs perform different functions in these animals, it indicates
1. That same structure developed along with different directions due to adaptation to different needs
2. All of them have similar anatomical structures like humerus radius, ulna, carpals. metacarpals and phalanges in their forelimb
3. These structures are homologous and show divergent evolution
4. All of these
The most apparent change during the evolutionary history of Homo sapiens is traced in
1. Loss of body hair
2. Walking upright
3. Shortening of the jaws
4. The remarkable increase in brain size
Which cancerous condition is caused as a result of complications of AIDS or is an opportunistic disease in affected humans?
1. Raus sarcoma
2. Genital warts
3. Kaposi's sarcoma
4. All of these
Which of the following surgeries is best postponed until after the tenth year of age to prevent immunological deficiency in a child?
1. Tonsillectomy
2. Gastrectomy
3. Appendicectomy
4. Septoplasty
The amount of that can diffuse through the diffusion membrane per unit difference in partial pressure is much higher as compared to that of . This is because
1. Solubility coefficient of is higher
2. Solubility coefficient of is lesser
3. Amount of gases in blood is independent of partial pressures of the gases in the atmosphere
4. Arterial blood contains more than .
What Volume of carbon dioxide is carried as dissolved gas by 100ml of oxygenated blood?
1. nil
2. 2.4ml
3. 2.7 ml
4. 1.5ml
Microscopical examination of the pus reveals that it contains
1. Lymph, RBCs and dead bacteria
2. Necrotic tissue, leucocytes and RBCs
3. Necrotic tissue, dead neutrophils and dead macrophages and tissue fluid
4. Tissue fluid, dead RBC, dead bacteria and necrotic tissue and dead leukocytes
Which of the following is not a function or characteristic feature of spleen?
1. It filters the disintegrated red blood cells from the blood flowing through it
2. It acts as a reservoir, holding a large amount of blood
3. In human embryo it is also the site of formation of both red and white blood cells
4. If the spleen is removed from the adult human the number of WBC decrease
If a man is , then which of the following can occur?
a. Their first child will die
b. Their first child will survive
c. The second child will suffer with erythroblastosis foetalis
d. No child would be born after the first delivery
1. Only(A)
2. Only(B)
3. (B) & (C)
4. Only (D)
Bee wax is the natural product of honey bee secreted by
1. Abdominal glands of the queen
2. Abdominal glands of worker bees
3. Abdominal glands of drones
4. Salivary glands of nurses
One should use contraceptive methods
1. To prevent pregnancy
2. To delay or space pregnancy
3. For the maintenance of reproductive health
4. All of these
In genetic engineering, the antibiotic resistance gene are used
1. As selectable markers
2. To select healthy vector
3. As sequences from where the replication starts
4. To keep culture free of infection
Which of the flowing a direct method of gene transfer?
1. Plasmid-mediated
2. Cosmid mediated
3. Ti vector mediated
4. Chemical mediated genetic transformation in which certain chemicals like polyethylene glycol help to uptake the foreign DNA
The figure below shows three steps (A, B, C) of polymerase chain reaction (PCR) Select the correct identification together with what it represents
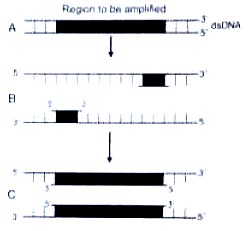
1. B - Denaturation at a temperature of about 98°C separating the two DNA strands
2. A - Denaturation at a temperature of about 50°C
3. C - Extension in the presence of heat-stable DNA polymerase
4. A - Annealing with two sets of primer
During the life cycle Fasciola hepatica infect its intermediate host and primary host at the following larval stages respectively?
1. Redia and Metacercaria
2. Cercaria and Redia
3. Metacercaria and Cercaria
4. Miracidium and metacercaria
The main objective of production of herbicide-resistant GM crops is to
1. Reduce herbicide accumulation in food articles for health safety
2. Eliminate weeds from the field without the use of manual labour
3. Eliminate weeds from the field without the use of herbicide
4. Encourage eco-friendly herbicides
The tubular epithelial cells in different segments of nephrons perform reabsorption either by active or passive transport. Which of the following substances in the filtrate are reabsorbed by passive transport?
1. Glucose and
2. Glucose and amino acids
3. Water and nitrogenous wastes
4. Amino acids and
Listed below are four abnormal conditions in urine (a-d) and the descriptions of the conditions
Column-I Column-II
(Abnormal condition) (Description)
a. Hematuria (i) Presence of hemoglobin in urine
b. Proteinuria (ii) Presence of pus cells in urine
c. Ketonuria (iii) Presence of blood cells in urine
d. Pyuria (iv) Presence of amino acids in urine
(v) Excess of protein level in urine
(vi) Presence of calculi
(vii) Presence of ketone bodies in urine
Which of the following are the correct matching of the two?
1. a(i), b(iv), c(vi), d(ii)
2. a(iii), b(iv), c(vi), d(ii)
3. a(iii), b(v), c(vi), d(ii)
4. a(iii), b(v), c(vii), d(ii)
Choose the incorrect match with respect to hormones and their functions
1. Parathyroid - Hypercalcemic hormone, it increases the blood calcium level by
hormone (PTH) increasing the bone resorption/demineralization
2. Glucocorticoids - Stimulates lipolysis, proteolysis, and gluconeogenesis and also stimulates R.B.C production
3. Atrial natriuretic - Cause vasodilation and decreases the blood pressure
factor
4. Somatostatin - Increases the release of growth hormone and thus increases the growth of the body
Out of the following statements for linkage/linkage group
a. The number of linkage group is equivalent to number of chromosomes of body cell.
b. Reduces the incidence of recombination
c. Useful for maintaining the good characters of the newly developed variety
d. Mother passes the alleles of a sex-linked traits to both sons and daughters
e. Females suffer from sex linked disorders more frequently than males
1. All are correct
2. Only e is correct
3. b, c and d are correct
4. a, d, e are wrong
In PNS, myelin sheath is formed by spiral wrapping of
1. Schwann cells
2. Process of oligodendrocytes
3. Microglia
4. Astrocytes
Which statement is correct with respect to single unit smooth muscles?
a. These have number of muscle fibres joined together closely.
b. These have number of muscle fibres not so closely joined.
c. All the fibres contract together as a single unit.
d. The individual fibres contract as separate units more or less as independent muscle fibre
1. a only
2. b & c
3. b & d
4. a & c
Fatty acids enter the aerobic respiratory pathway after being converted to the
1. Pyruvate
2. PGAL
3. Acetyl CoA
4. DHAP
Which of the following figure represents a typical placentation as seen in lemon?
1.
2.
3.
4.
In which region of the root, cells are very small, thin walled and with dense protoplasm :-
1. Region of elongation
2. Region of maturation
3. Region of meristematic
4. Both 2 and 3
Leaf base is swollen to form pulvinus in :-
1. Grasses
2. Some crucifers
3. Some cucurbits
4. Some leguminous plant
Given figure represents primary wall thickenings at corners. Which of the following wall materials is absent in primary wall thickening?
1. Pectin
2. Lignin
3. Cellulose
4. Hemicellulose
Given below 3 types of vascular bundles (A, B and C) are present, respectively in:-
1. Stem, root, leaf
2. Root, stem, leaf
3. Root, monocot stem and leaf, dicot stem
4. Monocot stem and leaf, dicot stem, monocot leaf
Match the column I, II and III and select the correct option given below:-
|
Column I |
Column II |
Column III |
|
A. Herbarium |
a. Living form |
(i) Quick referral system |
|
B. Museum |
b. Dead form |
(ii) Ex situ conservation |
|
C. Zoological Parks |
c. Dried form |
(iii) Set up in educational institutes |
|
D. Botanical Gardens |
(iv) Related with Food habit and behaviour |
1. A-b-i, C-c-i, B-b-ii, D-d-iv
2. B-b-iii, D-a-i, C-b-ii, A-c-iv
3. D-a-i, B-c-ii, C-a-iv, A-a-i
4. C-a-iv, A-c-i, B-b-iii, D-a-ii
Which one of the following statements about bryophytes is true?
1. Psilopsida is a class of bryophyta
2. Bryophytes are believed to be the most recently evolved group of plant.
3. The diploid generation of Bryophytes is unicellular.
4. True alternation of generations is present in bryophyta.
Meiosis occurs in Pteridophytes when:-
1. Gametes are formed
2. Prothallus are formed
3. Sex organs are formed
4. Spores are formed
The four sketch (A, B, C and D) given below represents four different types of organisms. Which one of these is correctly identified in the options given, along with its correct food and class :
A. B.
c.
D.
|
Name of organism |
Stored food |
Class |
|
a. Anabaena |
Glucose |
Slime mould |
|
b. Porphyra |
Floridean starch |
Red Algae |
|
c. Chara |
Laminarin |
Green Algae |
|
d. Chlamydomonas |
Manitol |
Brown Algae |
1. a
2. b
3. c
4. d
Fimbriae in bacteria are involved in
1. Conjugation
2. Attachment on host surface
3. Attachment on substratum
4. 2 and 3 both
Kingdom fungi is divided into various classes on the basis of :
1. Mode of nutrition
2. Pigments
3. Morphology of mycelium
4. Embryo
........(A)........ discovered viroids in ........(B)........
1. A = Linnaeus, B = 1753
2. A = M.W. Beijerinck, B = 1898
3. A = T.O. Diener, B = 1971
4. A = Stanley, B = 1935
The quasi fluid nature of the membrane is important from the point of view of function like :-
1. Cell division and cell growth
2. Endocytosis and secretion
3. Formation of intercellular junctions
4. All of the above
Given diagram showing flagellum's internal structure. In which of the following all the four parts labelled as A, B, C and D are correctly identified?
|
A |
B |
C |
D |
|
|
(1) |
Radial Spoke |
Central Microtubule |
Peripheral Microtubule |
Plasma Membrane |
|
(2) |
Bridge |
Plasma Membrane |
Central Sheath |
Peripheral Microtubule |
|
(3) |
Bridge |
Plasma Membrane |
Peripheral Microtubule |
Central Microtubule |
|
(4) |
Radical Spoke |
Plasma Membrane |
Central Microtubule |
Peripheral Microtubule |
1. (1)
2. (2)
3. (3)
4. (4)
During cell cycle, time taken for the completion of M-phase is :-
1. 5-10% of the total cell cycle
2. 10-20% of the total cell cycle
3. 30-40% of the total cell cycle
4. 40-60% of the total cell cycle
Following are the events occurs during meiosis:
(A) Appearance of chiasmata
(B) Synapsis
(C) Assembly of meiotic spindle
(D) Use of recombinase enzyme
Choose the correct sequence :-
1. A B C D
2. B D A C
3. D C B A
4. B C A D
Optimum temperature for C3 plant is :-
1. 20°C – 25°C
2. 30°C – 40°C
3. 40°C – 50°C
4. 0°C – 10°C
In light reaction water splitting complex in located:
1. With PSI, physically located on inner side of membrane of the thylakoid
2. With PSII, physically located on thylakoid membrane towards stroma
3. With PSI, physically located on thylakoid membrane towards stroma
4. With PSII physically located on inner side of the membrane of thylakoid
Which one of the following is incorrect regarding facilitated diffusion :-
1. Selective nature
2. Dependent on energy
3. Transport Saturation
4. Requirement of protein
Which one is responsible for inhibiting the growth of the lateral (axillary) buds, in plant :-
1. Auxin
2. Gibberllin
3. ABA
4. Ethylene
During aerobic respiration the amount of total ATP synthesised per kreb cycle will be :-
1. 8
2. 12
3. 6
4. 36
Which one of the following is oxidative pathway of nitrogen metabolism steps:-
1. Nitrate assimilation
2. Biological N2-fixation
3. Nitrification
4. Denitrification
Select incorrect statement regarding phytochrome pigment:-
1. It is a chromoprotein whose state is influenced by light
2. In darkness, PR is produced in cytoplasm
3. PFR is extraordinarily unstable
4. It is one of main structural protein pigment in plant kingdom
Which one of the following statments is incorrect ?
1. Himgiri variety of wheat is resistant to leaf and strip rust disease
2. Pusa swarnim 2 Variety of Bhindi has developed resistance against shoot and fruit borer insect pests
3. Pusa Gaurav Variety of Brassica is resistant to Aphids
4. High aspartic acid, low nitrogen and sugar content in maize leads to resistance to maize stem borers
Statins are produced by __________ and have been commercialised as __________ .
1. Bacteria, blood cholesterol lowering agent
2. Trichoderma, immunosuppressive agent
3. Yeast, blood cholesterol lowering agent
4. Bacteria, clot buster
Choose the odd one out with respect to parasitism
1. Parasites shows adaptations like loss of digestive system and high reproductive capacity.
2. Parasite may reduce the survival, growth and reproduction of host.
3. Life cycles of ectoparasites are more complex
4. Brood parasitism is seen in birds
Verhulst - Pearl logistic population growth is represented by
1.
2.
3.
4.
Mark the incorrect statement with respect to evil quartet
1. Habitat fragmentation reduces the area for organisms
2. Habitat loss is the major part of evil quartet
3. Parthenium argentatum is an exotic species and is harmful for ecosystem
4. Over exploitation resulted in extinction of water hyacinth from India
Using large outdoor plots, David Tilman confirmed that
1. Higher biodiversity leads to increase in productivity
2. High species diversity shows less year to year variation in total biomass
3. Role of human being in maintaining ecosystem
4. More than one option is correct
Allergic pollen grain belong to a group of plant species which are
1. Entomophilous
2. Anemophilous
3. Ornithophilous
4. Hydrophilous
Juvenile phase or vegetative phase is related with all, except
1. It is a pre-reproductive phase in the life cycle of an individual
2. It is a period of growth
3. It is of different durations in different organisms
4. It involves appearance of flowers in higher plants
What is the possible advantage if hybrids are made into apomicts?
1. Segregation of characters in the hybrid progeny
2. Farmers can keep on using the hybrid seeds to raise new crop in first year only
3. Absence of segregation in the hybrid progeny
4. More than one option is correct
Which of the following statement is correct for angiospermic endosperms?
1. Cannot be diploid
2. Always triploid
3. Nutritive tissue always
4. Cannot be absent
Which one of the following is incorrect with respect to lac operon?
1. It is inducible operon which controls anabolic pathway
2. Glucose or galactose cannot act as inducer
3. The repressor is synthesised constitutively from the i-gene
4. Repressor is active which binds to the operator region
In HGP, the fragments of DNA were sequenced using automated DNA sequencers. It worked on the principles of a method developed by
1. E.M. Southern
2. Frederick Sanger
3. Craig Venter
4. Wellcone Trust (U.K.)
If a hybrid DNA molecule labelled with is allowed to replicate four times in culture medium having , the percentage of hybrid DNA after third generation would be
1. 50%
2. 25%
3. 12.5%
4. 6.5%
Choose the incorrect option with respect to transcription unit
1. Regulatory sequnces may be present at both upstream and downstream of the promoter region
2. Promoter is absent at the 5' end of the structural gene
3. DNA strand which does not code for anything is called non - coding strand.
4. Termination sequence is present at 3' end of the coding strand.
If plant AaBBCc is corssed with a plant aaBbCc, then what percentage ofprogenies will have the genotype AaBBcc?
1. 6.25%
2. 12.5%
3. 25%
4. 66.6%
Match the column correctly
|
Column I |
Column II |
|
|
a. |
Phenylketonuria |
Autosomal trisomy associated with mongolism |
|
b. |
Down’s syndrome |
Gynaecomastia |
|
c. |
Klinefelter’s syndrome |
Autosomal recessive trait associated with mental retardation |
|
d. |
Turner’s syndrome |
Sterile females with rudimentary ovaries |
1. a (iii), b (i), c (iv), d (ii)
2. a (ii), b (iii), c (i), d (iv)
3. a (ii), b (i), c (iii), d (iv)
4. a (iii), b (i), c (ii), d (iv)
Select the incorrect statement with respect to sex linked inheritance in humans
1. Males are more susceptible to X - linked recessive diseases
2. Y - chomosome of males move to male offsprings, the inherited traits are called holandric
3. Females can be normal, carrier or diseased for X - linked dominant trait
4. Criss cross inheritance is possible in colour blindness.
Morgan utilised the drosophilla as an experimental material for his investigation as
1. Female flies are distinguishable from male flies due to their small size.
2. It can complete its life cycle in 12 months
3. It can be grwon in complex nutrient medium only
4. Single mating produce large number of progeny
Choose the odd one out with respect to succession
1. Succession and evolution are parallel processes
2. In abandon farmlands, primary succession occurs
3. Involves changes at every trophic level
4. Generally shows a trend from xeric to mesic conditons
Select a incorrect statement regarding pyramids
1. These are proposed by Charles Elton
2. It does not explain the role of organisms present at more than two trophic levels
3. Decomposers are not given importance while preparing a pyramid
4. It is sometimes upright, if prepared for energy.
Choose the odd one out with respect to ozone depletion in the stratosphere
1. UV rays have the ability to both degrade as well as form ozone layer.
2. UV rays are depleting the good ozone in the troposphere
3. CFC's are distributing the balance of ozone equilibrium
4. UV-B rays causes inflammation of cornea called colour blindness.
All the buses of Dehli were coverted to run on CNG by the end of 2002 as
A. It is cheaper than petrol or diesel
B. It burns more efficiently
C. It can easily be siphoned off
1. All are correct
2. Both A and C are correct
3. Only C is incorrect
4. Both B and C are correct

The IUPAC name of the above mentioned compound is -
1. 1,2,3-Tricyanopropane
2. Propane-1,2,3-tricarbonitrile
3. 1,2,3-Cyanopropane
4. Propane Tricarbylamine
How many stereoisomers does this molecule have?
CH3CH=CHCH2CHBrCH3
1. 4
2. 6
3. 8
4. 2
Equivalent conductances of NaCl, HCl and C2H5COONa at infinite dilution are 126.45, 426.16 and 91 . The equivalent conductance of C2H5COOH is :
1. 201.28
2. 390.71
3. 698.28
4. 540.48
Density ratio of O2 and H2 is 16 : 1. The ratio of their r.m.s. velocities will be-
1. 4 : 1
2. 1 : 16
3. 1 : 4
4. 16 : 1
The concentration of the Ag+ ions in a saturated solution of Ag2C2O4 is 2.2 x 10-4 M. The solubility product of Ag2C2O4 is
1. 2.42 x 10-8
2. 2.66 x 10-12
3. 4.5 x 10-11
4. 5.3 x 10-12
A solution containing 3.3 g of a substance in 125 g of benzene (b.p. 80°C) boils at 80.66°C. If Kb for one litre of benzene is 3.28°C, the molecular weight of the substance shall be
1. 127.20
2. 131.20
3. 137.12
4. 142.72
30 ml of 0.06 M solution of the protonated form of an anion acid methionine (H2A+) is treated with 0.09 M NaOH. Calculate pH after addition of 20 ml of base.[ pKa1, = 2.28 and pKa2 = 9.2.]
1. 5.5
2. 5.74
3. 9.5
4. None of the above
Sulphur burns according to the reaction
What volume of air, at 1 atm and 273 K, containing 21% oxygen by volume is required to completely burn sulphur present in 200 g of the sample?
(This sample contains 20% inert material which does not burn)
1. 23.52 litre
2. 320 litre
3. 112 litre
4. 533.33 litre
The quantity that does not change for a sample of a gas in a sealed tight container when it is cooled from at constant volume is-
1. Average energy of the molecule
2. Pressure of the gas
3. Density of the gas
4. Average speed of the molecules
The maximum entropy of mixing occurs when hexane and heptane are mixed respectively in the proportion of -
1. 8.6 g and 10.0 g
2. 8.6 g and 8.6 g
3. 10.0 g and 8.6 g
4. 10.0 g and 10.0 g
Given the reaction:

Which of the following statements is correct?
1. (b) has a lesser stability
2. (b) is more volatile than (a)
3. (a) is more volatile than (b)
4. (a) forms higher yields at a lower temperature
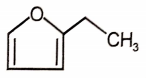
Which of the following will undergo Cannizzaro reaction?
1. 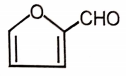 2.
2. 
3. 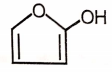 4.
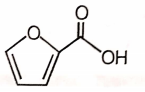
4. 
The product in the below mentioned reaction is
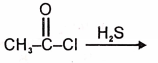
| 1. |  |
2. | 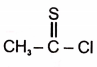 |
| 3. | 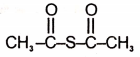 |
4. |  |
In a process, n-propyl chloride Is reacted with sodium butanoate in an aqueous medium. Alter the reaction diethyl ether is added and the solution Is shaken. The two layers are separated. The Incorrect statement with respect to this procedure
1. The reaction gives a solid product which precipitates in the aqueous solution.
2. The reaction takes place in the aqueous medium.
3. The product Is extracted in diethyl ether and the organic layer forms the upper layer.
4. The salt formed in the reaction remains In aqueous medium.
The correct order of the leaving group ability is:
| 1. | \(\text{ OCOC}_2\text{H}_5~\)>\(\text{OC}_2\text{H}_5\) >\(\text {OSO}_2\text{Me}\) > \(\text {OSO}_2\text{CF}_3\) |
| 2. | \(\text{OC}_2\text{H}_5\) > \(\text{ OCOC}_2\text{H}_5~\)> \(\text {OSO}_2\text{CF}_3\) > \(\text {OSO}_2\text{Me}\) |
| 3. | \(\text {OSO}_2\text{CF}_3\) > \(\text {OSO}_2\text{Me}\) > \(\text{ OCOC}_2\text{H}_5~\) > \(\text{OC}_2\text{H}_5\) |
| 4. | \(\text{ OCOC}_2\text{H}_5~\)>\(\text {OSO}_2\text{CF}_3\) > \(\text{OC}_2\text{H}_5\) > \(\text {OSO}_2\text{Me}\) |

1. Br2/KOH, H2/Pt, CH3COOH
2. P2O5/heat, H2/Pt, (CH3CO2)2O
3. (CH3CO)2O, H2/Pt, Br2/KOH
4. Br2/KOH, CH3COCl
The reaction of cyclohexanone with dimethylamine in the presence of the catalytic amount of an acid forms a compound, is generally known as
1. A Schiff s base
2. An enamine
3. An imine
4. An amine
The oxidation number of S in is:
1. +2
2. +4
3. +6
4. +7
If —D—glucose is dissolved in water and kept for a few hours, the major constituent(s) present in the solution is/are:
| 1. | α —D—glucose. |
| 2. | The mixture of ß—D-glucose and open-chain D-glucose. |
| 3. | Open chain D-glucose. |
| 4. | The mixture of α —D—glucose and ß—D-glucose. |
A bottle of solution contains 70% (w/w) acid. If the density of the solution is 1.54 g , the volume of the solution required to prepare 1 L of 1 N solution is
1. 90mL
2. 45 mL
3. 30 mL
4. 23 mL
The wavelength of the spectral line obtained in the spectrum of
when the transition takes place between levels whose sum is 4 and the difference is 2, will be:
1. 11.4 x cm
2. 1.14 x cm
3. 0.114 x cm
4. 1.14 x cm
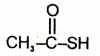
The structure of the monomer used for the preparation of plexiglass is
1. ![]()
2. 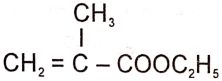
3. 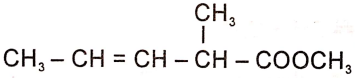
4. 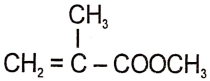
In a subshell, if the number of radial nodes is two times the number of angular nodes, then the minimum possible value of the principal quantum number (n) is:
[angular nodes are non-zero)
| 1. | 1 | 2. | 2 |
| 3. | 3 | 4. | 4 |
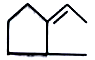
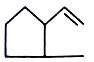
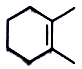
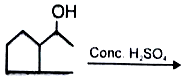 major product is
major product is
1. 
2. 
3. 
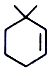
4. 
Which of the following statements is correct?
| a. | [Ni(H2O)6]2+ and [Ni(NH3)6]2+ have the different values of CFSE. |
| b. | [Ni(H2O)6]2+ and [Ni(NH3)6]2+ have the same value of the magnetic moment |
| c. | [Ni(H2O)6]2+ and [Ni(NH3)6]2+ have the same hybridization of nickel |
1. a & c only
2. b & c only
3. a & b only
4. All of these
All group-2 metals dissolve in liquid ammonia to produce a bright blue colour. The colour is due to
1. A change in the structure of ammonia
2. d-d transition
3. The spectrum of the solvate electrons
4. An electronic transition from a lower to a higher energy state
The ignition mixture in the aluminothermic process contains
1. Mg powder and BaO2
2. Mg powder, Al and BaO2
3. Mg and Al powder
4. Mg and Al oxide
When excess of KCN is added to a solution of CuSO4, the final product obtained is,
1. K2[Cu(CN)4]
2. K3[Cu(CN)4]
3. CuCN
4. K[Cu(CN)2]
Given lanthanoid ions ,the tendency to form the complexes is highest for:
1. Ce+3
2. Pm+2
3. Lu+3
4. Eu+2
The fusion of chromite ore (FeCr2O4) with Na2CO3 in air gives a yellow solution upon the addition of water. Subsequent treatment with H2SO4 produces an orange solution.
The yellow and orange colours, respectively, are due to the formation of:
1. Na2CrO4 and Na2Cr2O7
2. Cr(OH)3 and Na2Cr2O7
3. Cr2(CO3)3 and Fe2(SO4)3
4. Cr(OH)3 and Na2CrO4
Which of the following weighs less when weighed in magnetic field?
1. ScCl3
2. FeCl3
3. TiCl3
4. VCl3
The density and Eq. wt. of metal are 10.5 g and 100, respectively. The time required for a current of 3 amp to deposit a 0.005 mm thick layer of the same metal on an area of 80 is closest to
1. 120 s
2. 135 s
3. 67.5 s
4. 270 s
The equilibrium constant of a 2 electron redox reaction at 298 K is 3.8 x . The cell potential Eo (in V) and the free energy change ∆Go (in kJ mol-1 ) for this equilibrium respectively, are -
| 1. | -0.071, -13.8 | 2. | -0.071, 13.8 |
| 3. | 0.71, -13.8 | 4. | 0.071, -13.8 |
Consider the following Galvanic cell.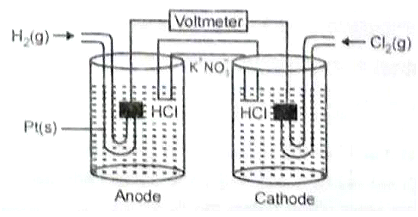
By what value the cell voltage change when concentration of ions in anodic and cathodic compartments both increased by a factor of 10 at 298
1. +0.0591
2. -0.0591
3. -0.1182
4. 0
The limiting molar conductivities at infinite dilution and are 239,125 and 128 S cm2/mole respectively. If a 0.1 M solution of has a molar conductivity of . The degree of dissociation is
1. 0.024
2. 0.24
3. 0.10
4. 0.05
The standard reduction potential for is 0.34 V.
The reduction potential at pH =14 for the above couple given is :
1. -0.22 V
2. +0.22 V
3. -0.34 V
4. +0.34 V
Calcium carbide with heavy water generates-
1.
2.
3.
4.
reacts with aqueous to give and Nal. The products of the reaction of Cl2 with aqueous are:
1. + NaCl
2. + HCl
3. + HCl
4. + NaCl
The 'S' atom among the following that does not have \(\mathrm{sp}^3\) hybridization is
| 1. | \(\mathrm{SO}^{2-}_4\) | 2. | \(\mathrm{SF}_4\) |
| 3. | \(\mathrm{SF}_2\) | 4. | \(\mathrm{S}_8\) |
What is the chemical formula of 'laughing gas'?
1. NO
2.
3.
4.
A colloid of Agl is formed by mixing AgNO3 with an excess of Kl. The electrolyte with the greatest coagulation value for it is:
1.
2. NaCl
3.
4.
In which series are the species listed in order of increasing size?
1. N, O, F
2. Na, Mg, K
3. Cr, Cr2+, Cr3+
4. Cl, Cl-, S2-
Which element from multiple bonds with itself and with carbon and oxygen?
1. P, As
2. N, As
3. N, P
4. N
The outermost electronic configuration of the most electronegative element is:
1.
2.
3.
4.
In a new system of units, mass is measured in multiples of 100 kg, length in multiples of 1 km, and time in multiples of 100 s. What is the value of 1 Joule in the new system of units?
(1)
(2)
(3)
(4)
The velocity of a particle moving along a straight line with constant acceleration \(a\) reduces to \(\frac{1}{5}\) of its initial velocity in time \(\tau.\) The total time taken by the body till its velocity becomes zero is:
1. \(\frac{4\tau}{3}\)
2. \(\frac{5\tau}{4}\)
3. \(\frac{4\tau}{5}\)
4. \(\frac{3\tau}{4}\)
A particle is thrown with a velocity 40 m/s. It passes through points A and B as shown in figure at time . The value of h is
(1) 15 m
(2) 10 m
(3) 30 m
(4) 20 m
Mark the correct statement
(1) The value of coefficient of friction is always smaller than 1
(2) A body slides on a surface, then force of friction always acts in the opposite direction of applied force
(3) Force of friction is zero for a block kept on an rough inclined plane
(4) The frictional force can never be more than the contact force on the body from the surface on which it is kept
What is the minimum value of force F such that at least one block leaves the ground in the given figure? (g=10 )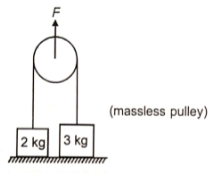
(1) 20 N, 2 kg leaves the ground first
(2) 30 N, 3 kg leaves the ground first
(3) 40 N, 2 kg leaves the ground first
(4) 50 N, 3 kg leaves the ground first
Block A moves on a smooth surface and collides with block B at rest. The maximum energy stored in the spring will be:
| 1. | \(\frac{1}{8} m v^2 \) | 2. | \(\frac{1}{4} \mathrm{~m} v^2 \) |
| 3. | \(\frac{1}{3} m v^2 \) | 4. | \(\frac{1}{2} m v^2\) |
If work done by string on block A is W, shown in the given arrangement, then the work done by the string on block B is:
1. - W
2.
3.
4.
A particle of mass makes an elastic one-dimensional collision with another stationary particle of mass . The fraction of kinetic energy of carried away by is-
1.
2.
3.
4.
A satellite of mass m at rest on the surface of Earth is to be launched into the parking orbit of radius 7R. The energy required for this process is [R-radius of Earth, g-acceleration due to gravity on Earth's surface]
(1)
(2)
(3)
(4)
A ball of mass 1 kg is thrown horizontally from the top of a tall tower with velocity m/s. The change in gravitational potential energy of the ball-earth system in 4 s will be
(1) 800 J
(2) 320 J
(3) 160 J
(4) 200 J
Aluminium and iron rods of same length and diameter are combined together as shown in figure. Force F is applied at one of the ends. The combined length is increased by 2 cm. The rods will have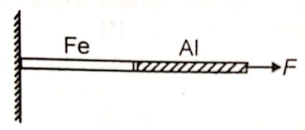
(1) Same stress and strain
(2) Different stress and strain
(3) Same stress and different strain
(4) Same strain and different stress
A solid is suspended from an independent support in a liquid placed on a weighing machine, due to which the weight of the liquid:
| 1. | Increases |
| 2. | Decreases |
| 3. | Remain unchanged |
| 4. | May increase or decrease |
Water flows through a frictionless tube with a varying cross-section as shown in figure. Variation of pressure P at a point along axis at distance x from left end is roughly given by-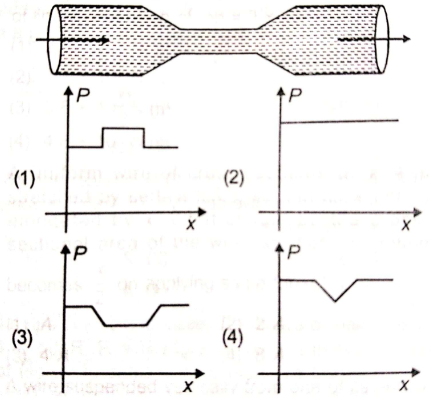
| 1. | \(2v_0 \over \sqrt{3}\) | 2. | \(\sqrt{2}v_0 \over 3\) |
| 3. | \({2 \over 3}v_0\) | 4. | \(\sqrt{\frac{2}{3}}v_0\) |
The time period of a simple pendulum in a stationary lift is T. If the lift moves upwards with an acceleration g, then the new time period will be:
1. Infinite
2.
3.
4. 0.707 T
A sound source emitting the sound moves towards a stationary observer with speed 30 m/s. The frequency perceived by the observer is increased by (speed of sound = 330 m/s)
(1) 5%
(2) 10%
(3) 12.5 %
(4) 15%
| 1. | \(100~\text{Hz}\) | 2. | \(200~\text{Hz}\) |
| 3. | \(50~\text{Hz}\) | 4. | \(400~\text{Hz}\) |
A faulty thermometer reads freezing point and boiling point of water as respectively, the correct value of temperature as it reads is
(1)
(2)
(3)
(4)
Which of the following processes will take greatest time?
(1) Cooling down of water from
(2) Cooling down of water from
(3) Cooling down of water from
(4) All will take equal time
The thermodynamic state of an ideal gas is varied through different processes as shown in the figure. The change in internal energy of the process A, B and C respectively will be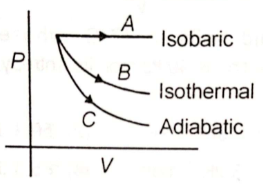
(1) +, -, +
(2) 0, +, -
(3) +, 0, -
(4) -, +, 0
At constant pressure, the ratio of increase in volume of an ideal gas per unit rise in kelvin temperature to its original volume is (T=absolute temperature of gas)
(1)
(2)
(3)
(4) T
An electric dipole is in unstable equilibrium in external uniform electric field. Angle between its dipole moment and the external electric field is
(1)
(2)
(3)
(4)
Three identical metallic plates with area A are kept as shown. Plate 1 is given -5Q charge and plate 3 is given Q charge. Electric field intensity at point x is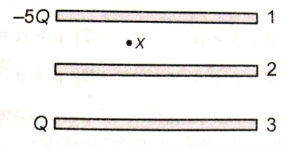
(1)
(2)
(3)
(4)
How much charge is supplied by the battery when the switch is closed in the circuit?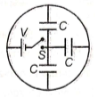
(1)
(2) CV
(3)
(4) 3 CV
Two concentric metallic spherical shells A and B of radii a and b respectively (b>a) are arranged such that outer shell is earthed and inner shell is charged to Q. Charge on the outer surface of outer shell will be
(1)
(2)
(3) -Q
(4) Zero
Two concentric and coplanar circular loops of radii are placed as shown in figure. If current I is flowing in the bigger loop, then magnetic flux linked with smaller loop is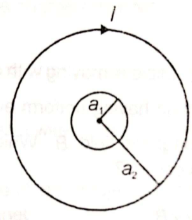
(1)
(2)
(3)
(4)
On connecting a shunt of 10 , the deflection in a moving coil galvanometer falls from 40 divisions to 6 divisions. The resistance of the galvanometer is
(1)
(2)
(3)
(4)
Magnetic induction at an axial point of a short magnet at a distance r from the centre of dipole is . Its value at the equatorial point of the short magnet at same distance from centre of dipole is
(1)
(2)
(3)
(4)
Charged pollen grains are lying on a frictionless table. These are now subjected to certain field and are found to be moving by these fields. The field cannot be
1. Time dependent electric field
2. Position dependent electric field
3. Time dependent magnetic field
4. Position dependent magnetic field
The figure shows a parallel plate capacitor being charged by a battery. If \(X\) and \(Y\) are two closed curves then during charging, \(\oint \vec{B}.d\vec{l}\) is zero along the curve:

1. \(X\) only
2. \(Y\) only
3. Both \(X\) & \(Y\)
4. Neither \(X\) nor \(Y\)
A capacitor and a resistor are connected in series across the AC supply. Which of the following phasor diagrams may be correct?
| 1. |  |
2. |  |
| 3. |  |
4. |  |

If half life of a radioactive substance is 10 hours, then its mean life is
(1) 14.4 h
(2) 7.2 h
(3) 20 h
(4) 6.93 h
A sample of radioactive material has initial mass A, decay constant B, molecular weight C and Avogadro's constant D. The activity of the sample after time t will be
(1)
(2)
(3)
(4)
An electron in an atom makes transition from the energy state . The wavelength of the emitted photon is
(1)
(2)
(3)
(4)
If 5th order maxima of wavelength 4000 in Young's double slit experiment coincides with nth order maxima of wavelength 5000 , then n is equal to
(1) 5
(2) 8
(3) 4
(4) 10
The graph between resolving power P and accelerating potential V for an electron microscope is-
A particle of mass m moves along the line y=x with speed v. The magnitude of angular momentum about the point is
(1)
(2) 2mva
(3) mva
(4)
The moment of inertia of a uniform semicircular disc of mass M and radius R about axis through centre of mass on its plane as shown in figure is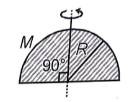
(1)
(2)
(3)
(4)
If switch S is closed, then the reading of ammeter will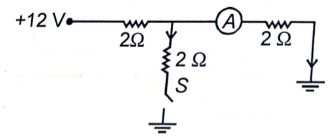
(1) Decrease by 1 A
(2) Remain same
(3) Increase by 1 A
(4) Increase by 4 A
Figure shows the variation of photoelectric current (i) with voltage (V) between the electrodes in a photocell for two different radiations. If are the intensities of the incident radiations and are their respective frequencies, then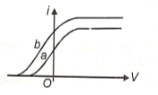
(1)
(2)
(3)
(4)
A n-p-n transistor operates in a common emitter mode as shown. Given that . The value of is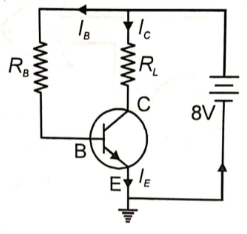
(1)
(2) 2
(3) 3
(4) 4
The combination of gates shown in diagram is equivalent to
(1) OR
(2) AND
(3) NAND
(4) XOR
An object is placed at a distance of 20 cm on the left side of a thin lens as shown in the figure. The points denote centres of curvature. The magnification produced by the lens is (Refractive index of glass is 1.5)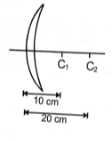
(1) -2
(2) +2
(3) -1/2
(4) +1/2